Anatomical Anterior Cruciate Ligament Reconstruction: A Rationale
Anterior cruciate ligament injuries occur frequently in young and active persons.
ABSTRACT: Patients with anterior cruciate ligament injuries typically need operative reconstruction to restore normal knee biomechanics and prevent the early development of osteoarthritis. Conventional nonanatomical reconstructive techniques have been ineffective. True anatomical reconstruction may be achieved only by customizing each procedure on the basis of an individual patient’s native anatomy. A comprehensive history and physical examination, including an assessment of the rupture pattern, help preoperative planning. The physical examination should assess knee function, range of motion, swelling, laxity, and pain and include tests to rule out concomitant injuries and neurovascular involvement. Imaging with conventional radiography, MRI, and other modalities plays a definitive role in diagnosis. The main priorities of rehabilitation are regaining range of motion and quadriceps strength.
Anterior cruciate ligament (ACL) injuries occur frequently in young and active persons. Patients typically have a need for operative reconstruction of the ligament to restore normal knee biomechanics and, subsequently, prevent the early development of osteoarthritis (OA) of the injured knee. However, conventional nonanatomical reconstructive techniques have been ineffective in restoring rotational stability and in preventing early degenerative changes of the articular cartilage.
To restore knee biomechanics and function adequately, returning the anatomy to its native situation as closely as possible is of paramount importance. Objectifying anatomy by taking measurements helps determine surgical technique. Surgery should be individualized to each patient, and nonanatomical techniques should be eliminated because they are a risk factor for the development of early OA. In this article, we provide a concise rationale for our evaluation and treatment of patients who have a torn ACL.
BACKGROUND
The ACL is one of the main knee stabilizers; specifically, it controls anteroposterior (AP) and rotational stability. As such, this ligament is prone to tearing in sports that involve cutting and pivoting, and ACL injury has become one of the most common sports injuries in an increasingly young and active population. The incidence rate for ACL rupture is 35 of 100,000 persons; the risk of injury is 2 to 3 times higher for females.1 Because the ACL does not have the potential to heal adequately when torn, reconstructive surgery generally is the treatment of choice.
FIGURE 1
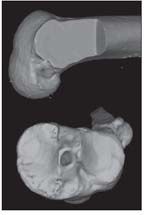
A 3-dimensional CT scan shows a nonanatomical single-bundle (SB) anterior cruciate ligament reconstruction and demonstrates a posterolateral (PL) tibial tunnel and a “high” anteromedial (AM) femoral tunnel.
Although the ACL has been acknowledged as consisting of 2 distinct functional bundles-anteromedial (AM) and posterolateral (PL)-the ligament has long been reconstructed nonanatomically with a single graft (Figure 1).2 The conventional nonanatomical single-bundle (SB) technique has shown good results in the short term, with improvement in subjective knee stability.3 At long-term follow-up, however, nonanatomical SB ACL reconstruction (ACL-R) does not restore native knee biomechanics and the early onset of OA seems to occur at the same rate as in knees that have not undergone surgery.4-7
In addition, subjective knee instability may persist, leaving patients unable to return to their previous activity level. In a study conducted by Ardern and associates,8 only 45% of patients had returned to their previous level of activity at 2 to 7 years after SB ACL-R.
Anatomical double-bundle (DB) ACL-R, an alternative to SB ACL-R, is suggested to more closely reproduce the native anatomy of the ACL by restoring both the AM and the PL bundles individually.9 In cadaver studies, anatomical DB ACL-R demonstrated better restoration of biomechanical properties of the knee, especially rotational stability, than did nonanatomical SB ACL-R.10
Hussein and colleagues11 conducted a study (level of evidence, I) that demonstrated the clinical superiority of anatomical ACL-R. At 3- to 5-year follow-up with an 85% follow-up rate, both AP and rotational knee stability were better restored with an anatomical DB or SB ACL-R than with a nonanatomical SB ACL-R.
True anatomical ACL-R may be achieved only by customizing each procedure on the basis of an individual patient’s native anatomy. Only then can the ACL be functionally restored to its native dimensions, collagen orientation, and insertion sites.12
PATIENT EVALUATION
Clinical Assessment
Patients who have a torn ACL typically present with an unstable, swollen, and painful knee. They may complain of difficulty in bearing weight and diminished range of motion. Swelling typically decreases and, consequently, range of motion improves within the first weeks after the injury. Patients with chronic ACL deficiency may have less evident complaints and experience only a nonspecific pain and recurrent instability of the knee. For the greatest potential for a successful outcome, surgery should be performed when swelling has diminished and range of motion is near normal.13
A comprehensive history and physical examination, including an assessment of the rupture pattern and the presence of concomitant injuries, help preoperative planning. The physical examination should assess knee function, range of motion, swelling, laxity, and pain. The examination also should include tests to rule out any concomitant injuries and neurovascular involvement. Specific assessments include the anterior and posterior drawer, Lachman, pivot shift, varus/valgus stress, McMurray, and dial tests. AP instability may be quantified by KT-1000 arthrometer testing.
FIGURE 2
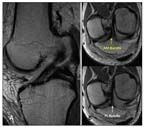
Shown are special anterior cruciate ligament (ACL) sequences on MRI in which the slices are parallel to the plane of the ACL
Imaging
FIGURE 3
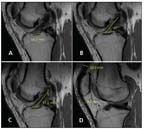
Preoperative measurements for anterior cruciate ligament (ACL) surgery made on standard sagittal MRI include tibial insertion site length (A)
Imaging with conventional radiography, MRI, and other modalities plays a definitive role in diagnosis and helps preoperative planning of individualized surgery. Special oblique MRI planes obtained in alignment with the ACL allow for a more complete evaluation of the two bundles (Figure 2).14 Preoperative measurements include tibial insertion site length, ACL length, ACL inclination angle, and quadriceps and patellar tendon thickness on standard sagittal MRI. These measurements are critical to the individualization process, and they help determine reconstruction technique and graft choice (Figure 3).
FIGURE 4

For anterior cruciate ligament surgery, the patient lies in the supine position with the operative leg in the leg holder and the nonoperative leg in the lithotomy position (A). The operative knee should allow for 120° of flexion (B) and full extension (C).
SURGICAL TECHNIQUE
Once anesthesia is induced, the physical examination is repeated to evaluate ligamentous knee function more specifically. The patient is placed in the supine position. A tourniquet is placed high on the operative extremity, which is fixed in a leg holder allowing for at least 120° of knee flexion. The contralateral leg is positioned in the lithotomy position outside of the surgical field (Figure 4).
Portal Placement and Arthroscopy
A high anterolateral portal (ALP) is created above the fat pad to allow for an unobstructed view of the notch and the tibial ACL insertion site. At this point, diagnostic arthroscopy of the knee is performed.
FIGURE 5
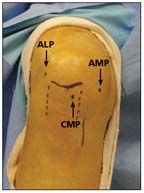
Approximation of portals is shown here. The anterolateral portal (ALP) is placed high, above the fat pad.
Next, with a spinal needle under arthroscopic visualization, the central medial portal (CMP) and accessory medial portal (AMP) are placed as described by Araujo and associates (Figure 5).15 The CMP provides an overview of the intercondylar notch and the femoral ACL insertion site, which is critical for evaluating the rupture pattern and appreciating the native anatomy. The AMP is used mainly as a working portal.
Intraoperative measurements of the insertion sites and notch size are obtained with an arthroscopic ruler (Figures 6 and 7). On the basis of both the preoperative and intra-operative measurements, the surgeon makes a final decision on surgical technique and graft choice.
A tibial insertion site length of less than 14 mm or a narrow notch (less than 12 mm wide) technically precludes DB ACL-R. As such, an SB technique would be preferred. Insertion site lengths between 14 and 18 mm permit the SB and the DB techniques. When the tibial insertion site is longer than 18 mm, a DB procedure is preferred.
FIGURE 6
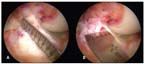
At the tibial insertion site, the site length (A), midwidth (B), and individual bundle widths are measured with an arthroscopic ruler.
Anatomical Double-Bundle Reconstruction
The notch and ACL remnants are visualized through the CMP and carefully dissected with a thermal device to identify the AM and PL insertion sites and their bony margins. The scope is then switched to the AMP to visualize and measure the femoral insertion site (Figure 8).
FIGURE 7
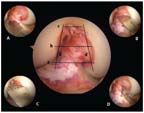
The notch width is measured at 3 levels: the base (A), the middle (B), and the apex (C). Subsequently, the notch height is measured on the lateral and medial wall of the notch (D).
A DB procedure may be performed with soft tissue grafts or a split quadriceps graft with a bone block (Figure 9). For soft tissue grafts, 2 femoral tunnels should be placed in the center of the AM or PL insertion site. However, if a quadriceps graft is used, a single femoral tunnel positioned midway between the AM and PL insertion sites is preferred to accommodate the bone block.
FIGURE 8
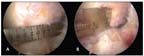
With an arthroscopic ruler, the femoral insertion site length (A), midwidth (B), and individual bundle widths are measured.
A pointed awl is used to create the initial hole in the PL bundle insertion site center for the first tunnel, and a guidewire is advanced. The femoral PL tunnel is then drilled over this guidewire through the AMP. The knee is flexed to 120° to avoid iatrogenic damage and to obtain maximum tunnel length. The drill diameter should be 1 mm smaller than the desired tunnel size. The tunnel is drilled to a depth of at least 20 mm. A suspensory device drill is used to breach the far lateral cortex. The total length of the tunnel is measured, and the tunnel is then drilled by hand to an adequate depth for suspensory device fixation. The desired diameter is achieved with the use of a hand dilator.
Attention is then turned to the tibial side. A 3- to 4-mm vertical incision is made in the proximal AM aspect of the leg between the anterior tibial tubercle and the medial tibial crest. This incision is used for tibial AM and PL tunnel drilling. With the scope through the ALP, the tibial insertion site is dissected carefully with a thermal device and the PL and AM insertion sites are marked. A tip-to-tip tibial aiming guide set to 45° is then placed in the marked PL insertion site.
FIGURE 9

For split quadriceps autograft with bone block, the purple arm is placed in the posterolateral position and the other arm in the anteromedial position. The suspensory device is already mounted on the bone block.
A guidewire is drilled through the aiming device (Figure 10), which is then reset to 55° for AM tunnel placement through the CMP following the same steps. To ensure adequate bone bridge between the tunnels, the entrances should be 1 cm apart on the leg and intra-articular pin spread should be measured. The PL tunnel is drilled first and advanced over the guidewire. Desired tunnel diameter (1 mm larger than the drill size) is achieved subsequently with a hand dilator. The same steps apply to AM tunnel drilling.
FIGURE 10
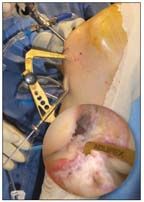
A tip-to-tip tibial aiming guide is placed in the center of the marked posterolateral (PL) and anteromedial (AM) insertion sites.
The AM femoral tunnel can be drilled through the AM tunnel (100% of cases) or through the PL tibial tunnel (about 60% of cases). If the transtibial approach allows for an anatomical placement of the AM tunnel, this method should be used. However, it necessitates a half moon drill bit, because the tibial PL tunnel has a smaller diameter than the would-be femoral AM tunnel.
If an anatomical position is not feasible with the transtibial approach, AMP drilling is required. The femoral AM tunnel drilling follows the same steps as described for the femoral PL tunnel drilling.
Next, the grafts are passed. The PL graft is passed first and fixed on the femur with a suspensory device, followed by passing of the AM graft, which is fixed in similar fashion. The knee is then cycled several times with tension applied to the tibial ends of the grafts. On the tibial side, the PL bundle is fixed at 0° and the AM bundle at 45° of knee flexion, both with bioabsorbable interference screws.
Anatomical Single-Bundle Reconstruction
This reconstruction follows the same protocol as the DB procedure. However, it has the following differences: 1 femoral tunnel and 1 tibial tunnel positioned midway between the AM and PL insertion sites and graft fixation at 15° of knee flexion.
COMPLICATIONS
Anatomical SB and DB procedures have the same general complications, including wound infection, hemarthrosis, arthrofibrosis, effusion, neurovascular injury, tunnel widening, tibial or femoral fractures, and deep venous thrombosis. The DB technique is more complex technically and therefore more prone to complications in inexperienced hands. Therefore, the DB concept should be first solidified in an SB approach before a DB reconstruction is attempted.
REHABILITATION
Protocols are the same for anatomical SB and DB ACL-R. The main priorities of rehabilitation are regaining range of motion and quadriceps strength.
For the first postoperative week, patients are kept in a hinged knee brace locked in full extension. Ambulation is permitted with crutches. At the end of week 1, continuous passive motion is initiated and progresses to full extension.
Patients typically are weaned from braces and crutches at about 6 weeks, depending on quadriceps muscle strength. At this point, straight-line walking can begin, with gradual progression to straight-line jogging and stationary biking at about 3 months. Pivoting and cutting generally are permitted at about 6 months, with a gradual return to sports activity at about 9 to 12 months (for both autografts and allografts).
Patients’ progress through the rehabilitation phases varies with their “readiness” as assessed by the physical therapist and the operating surgeon’s discretion. Readiness has subjective and objective components, including performance on rehabilitation tests, clinical findings, and even MRI evaluation to assess graft healing.
SUMMARY AND CONCLUSIONS
ACL injuries are common in an increasingly young and active population. Diagnosis is based on a combination of the patient’s history and complaints, physical examination results, and MRI and arthroscopic evaluation findings.
Conventional surgical techniques do not account for individual anatomy, which may lead to nonanatomical placement of the graft, subsequent altered knee biomechanics and, eventually, an early onset of OA. Individualized anatomical ACL-R restores the ACL to its native situation with respect to individual patient characteristics. Individualized surgery and a rehabilitation protocol that considers both patient progression and graft healing provide the patient with great potential for a successful outcome.
References:
REFERENCES
1. Gianotti SM, Marshall SW, Hume PA, Bunt L. Incidence of anterior cruciate ligament injury and other knee ligament injuries: a national population-based study. J Sci Med Sport. 2009;12:622-627.
2. Palmer I. On the injuries to the ligaments of the knee joint: a clinical study: 1938. Clin Orthop Relat Res. 2007;454:17-22.
3. Lewis PB, Parameswaran AD, Rue JP, Bach BR Jr. Systematic review of single-bundle anterior cruciate ligament reconstruction outcomes: a baseline assessment for consideration of double-bundle techniques. Am J Sports Med. 2008;36:2028-2036.
4. Tashman S, Collon D, Anderson K, et al. Abnormal rotational knee motion during running after anterior cruciate ligament reconstruction. Am J Sports Med. 2004;32:975-983.
5. Lohmander LS, Ostenberg A, Englund M, Roos H. High prevalence of knee osteoarthritis, pain, and functional limitations in female soccer players twelve years after anterior cruciate ligament injury. Arthritis Rheum. 2004;50:3145-3152.
6. Gillquist J, Messner K. Anterior cruciate ligament reconstruction and the long-term incidence of gonarthrosis. Sports Med. 1999;27:143-156.
7. Daniel DM, Stone ML, Dobson BE, et al. Fate of the ACL-injured patient: a prospective outcome study. Am J Sports Med. 1994;22:632-644.
8. Ardern CL, Taylor NF, Feller JA, Webster KE. Return-to-sport outcomes at 2 to 7 years after anterior cruciate ligament reconstruction surgery. Am J Sports Med. 2012;40:41-48.
9. Odensten M, Gillquist J. Functional anatomy of the anterior cruciate ligament and a rationale for reconstruction. J Bone Joint Surg. 1985;67A:257-262.
10. Yagi M, Wong EK, Kanamori A, et al. Biomechanical analysis of an anatomic anterior cruciate ligament reconstruction. Am J Sports Med. 2002;30:660-666.
11. Hussein M, van Eck CF, Cretnik A, et al. Prospective randomized clinical evaluation of conventional single-bundle, anatomic single-bundle, and anatomic double-bundle anterior cruciate ligament reconstruction: 281 cases with 3- to 5-year follow-up. Am J Sports Med. 2012;40:512-520.
12. Yamamoto Y, Hsu WH, Woo SL, et al. Knee stability and graft function after anterior cruciate ligament reconstruction: a comparison of a lateral and an anatomical femoral tunnel placement. Am J Sports Med. 2004; 32:1825-1832.
13. Mauro CS, Irrgang JJ, Williams BA, Harner CD. Loss of extension following anterior cruciate ligament reconstruction: analysis of incidence and etiology using IKDC criteria. Arthroscopy. 2008;24:146-153.
14. Steckel H, Vadala G, Davis D, et al. 3-T MR imaging of partial ACL tears: a cadaver study. Knee Surg Sports Traumatol Arthrosc. 2007;15:1066-1071.
15. Araujo PH, van Eck CF, Macalena JA, Fu FH. Advances in the three-portal technique for anatomical single- or double-bundle ACL reconstruction. Knee Surg Sports Traumatol Arthrosc. 2011;19:1239-1242.