Fighting fibromyalgia: Five key issues
ABSTRACT: Physicians treating patients with fibromyalgia syndrome(FMS) should rule out coexisting disorders and establish possible inducingfactors. They can work with patients to address their symptomsby providing patient education, instilling a sense of self-worth,advising avoidance of disability and narcotic medications, offeringinstruction in sleep hygiene, establishing anxiety reduction measures,and recommending an exercise program. Patients who havespecial needs and refractory cases may benefit from referral to amusculoskeletal specialist. Physicians can best improve patients'prognosis and quality of life by addressing several issues: whetherthe FMS is primary or secondary, whether there are underlying psychosocialstressors, whether pain is regional or widespread, how toapproach exercise for therapy, and determining which medicationsto use. (J Musculoskel Med. 2008;25:172-184)
ABSTRACT: Physicians treating patients with fibromyalgia syndrome (FMS) should rule out coexisting disorders and establish possible inducing factors. They can work with patients to address their symptoms by providing patient education, instilling a sense of self-worth, advising avoidance of disability and narcotic medications, offering instruction in sleep hygiene, establishing anxiety reduction measures, and recommending an exercise program. Patients who have special needs and refractory cases may benefit from referral to a musculoskeletal specialist. Physicians can best improve patients' prognosis and quality of life by addressing several issues: whether the FMS is primary or secondary, whether there are underlying psychosocial stressors, whether pain is regional or widespread, how to approach exercise for therapy, and determining which medications to use. (J Musculoskel Med. 2008;25:172-184)
Primary care physicians are the first line of triage for patients who have fibromyalgia syndrome (FMS). They are responsible for ruling out coexisting disorders and establishing possible inducing factors that may warrant specific management. Once these goals are accomplished, they can steer patients in a positive direction toward addressing their symptoms without becoming stigmatized. Providing patient education, instilling a sense of self-worth, advising avoidance of disability and narcotic medications, offering instruction in optimal sleep hygiene, establishing anxiety reduction measures, and recommending a comprehensive exercise and stretching program all may help this effort. Involving patients in management decisions has the potential to improve physician-patient interaction and, as a result, improve outcomes.1
Patients with early FMS that is diagnosed and managed in the primary care setting have a very good prognosis; more than half no longer meet the American College of Rheumatology criteria within 2 years.2 Those who have special needs (eg, counseling and local injections) and refractory cases may benefit from referral to a musculoskeletal specialist (eg, rheumatologist, neurologist, orthopedist, osteopath, or physical medicine and rehabilitation physician) to confirm the diagnosis in a single encounter or work with the primary care physician to treat the patient. Long-standing patients with FMS who have psychological issues (eg, bipolar illness or posttraumatic stress disorder [PTSD]) have a poorer prognosis, but the family physician or internist should remain the "captain of the ship" in directing treatment.
Primary care physicians can best manage FMS, and thereby improve patients' prognosis and quality of life, by addressing 5 key issues. In this article, we describe several cases of FMS that demonstrate the issues that frequently arise and how primary care physicians can address them with improved communication techniques to improve outcomes.
BASIC SCIENCE
FMS is a syndrome, or group of symptoms that occur together, rather than a disease. It is characterized by chronic widespread musculoskeletal pain of at least 3 months' duration in all 4 quadrants of the body and is thought to be a "central sensitization syndrome"3 (afferent sensory inputs into the dorsal root ganglion of the spinal column overwhelm the "gated"protective mechanism). A "windup" phenomenon (hyperexcitability with a low discharge threshold that worsens with each sensory input) creates greater discomfort than is seen in most persons. Thin C unmyelinated fibers and large myelinated A fibers and autonomic B fibers carry signals that result in amplified pain, hypervigilance, and discomfort from sensations that most persons would find pleasurable (eg, gentle stroking). Other central sensitization syndromes include irritable bowel syndrome (IBS), irritable bladder syndrome, chronic pelvic pain, chronic fatigue syndrome, tension headache, and temporomandibular joint dysfunction syndrome.
The primary manifestations of FMS include altered sleep architecture, aching, and fatigue (Table 1). FMS affects 2% to 3% of the US population4; most patients are women, and the syndrome often develops during their reproductive years.
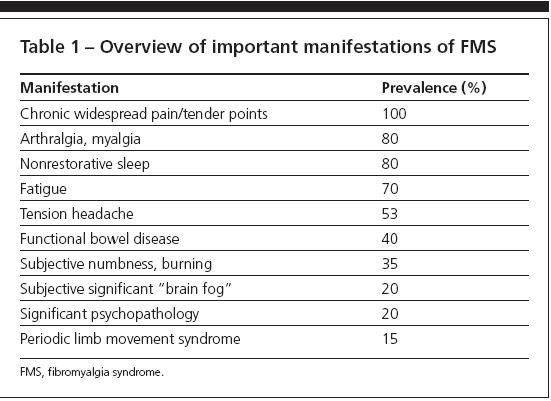
Table 1
Table

Table
CASES AND ISSUES
Clinical case scenario #1
A 36-year-old white woman with a past history of postpartum depression and hypothyroidism presents in a primary care physician's office with a complaint of pain all over her body. The symptoms appeared to start 3 months earlier after the patient slipped and fell while shopping in a department store. The fall aggravated the low back pain that had started during her pregnancy 18 months earlier. During the following week, the pain had spread from her lower back up her spine and into her legs. Her sleep became progressively disturbed,and the pain spread to her shoulders, neck, and arms.
The patient's current medications include fluoxetine, 20 mg/d, and levothyroxine, 100 μg/d. Results of laboratory tests performed within the last 3 months showed a normal complete blood cell count, erythrocyte sedimentation rate, and thyroid-stimulating hormone level; results for rheumatoid factor and antinuclear antibodies were negative.
Issue #1: Is FMS primary or secondary?
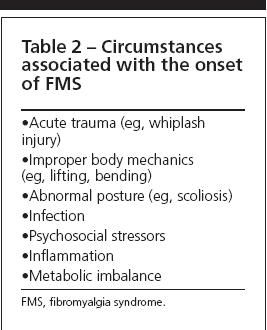
The many factors associated with the onset of FMS include motor vehicle accidents, infections, continuous work-related overuse of some muscles (eg, heavy lifting or pulling), and psychosocial stressors.5 In addition, well-defined medical conditions are known to involve an increased prevalence of FMS. Patients who have inflammatory disorders (eg, rheumatoid arthritis), metabolic disturbances (eg, hypothyroidism), or underlying malignancies frequently have a "reactive" FMS when undertreated inflammation or tissue insults result in muscle spasm. Chronic use of corticosteroids induces cutaneous hyperesthesia and an acute FMS like syndrome when the dose is reduced. In addition, some patients are told that they have FMS when there are other explanations for their symptoms (Tables 2 and 3).
When a diagnosis of FMS is made, the primary care physician may play an important role in assessing what other conditions are present and ruling out those that otherwise could stigmatize a patient given a diagnosis for a disorder that he or she does not have. The physician should explain to the patient that FMS is simply a set of symptoms and physical findings that can be nonspecific and provide important clues toward understanding why the patient feels unwell. Managing comorbid conditions goes a long way toward ameliorating FMS symptoms.
Clinical case scenario #1 (continued)
The patient admits to feeling upset because the pain prevents her from adequately taking care of her child and her home. She denies having feelings of hopelessness and episodes of crying that made her isolated and unable to care for her child in the early postpartum period. She believes that she is constantly behind in her housework and often blames herself for her condition. She fears that her symptoms are the result of an undiagnosed serious condition and that the tests performed so far have failed to pick it up.
Issue #2: Are there underlying psychosocial stressors?
Although many patients with FMS have a history of depression in their lifetime, only 15% to 20% are depressed at any given physician visit.6 Primary care physicians may be able to handle many FMS flares in a crisis intervention mode by addressing a patient's divorce, death of a loved one, or job loss.
The most common behavioral conditions associated with FMS are generalized anxiety disorder, PTSD, and mood disorders.6,7 Anxiety may be related to a patient's perception that he has a serious medical condition, psychosocial stressors, or discomfort at not feeling well. Anxious patients with FMS often have perfectionistic tendencies, tend to make lists, exhibit hypervigilance, and sleep poorly; however, they tend to hide their discomfort in social settings.
Anxious patients are the most responsive to FMS-related interventions, counseling, biofeedback, reassurance, cognitive-behavioral therapy, and complementary regimens that promote relaxation. Most remain under the care of their primary care physician, except for an occasional musculoskeletal specialist visit.
However, when PTSD is associated with a personal or family history of alcoholism, substance abuse, domestic violence, emotional or sexual abuse, or military service, simply injecting tender points and prescribing a tricyclic antidepressant and physical therapy has a very low improvement rate.The physician needs to query the patient in a sensitive but methodical way. Persons with such a history who have ongoing psychosocial stressors tend to cope poorly and usually benefit from ongoing counseling and long-term psychotropic interventions.
Mood disorders range from bipolar illness to alexithymia. Bipolar illness is 150 more times common in patients with FMS than in healthy controls and does not respond to most interventions.8 However, primary care physicians may have a significant impact on dysthymic states, catastrophizing, and depression by prescribing tricyclic antidepressants, selective serotonin reuptake inhibitors (SSRIs), duloxetine, benzodiazepines, or anticonvulsants (eg, pregabalin).
Patient expectations need to be addressed. For example, make sure that patients understand that pharmacotherapy is adjunctive to other interventions and that there are some things that the physician cannot do to help them.
Clinical case scenario #2
Mr Jones, a 52-year-old store owner, presents with a complaint of pain over the right shoulder blade and base of the neck on the right side that has lasted for 3 years. The pain appeared to be intermittent at its onset and to worsen with excessive bending or twisting of the neck and carrying of heavy weights in his right arm. One year before, the patient was evaluated for cervical radiculopathy with MRI of the cervical spine and electromyography (EMG).The results showed normal neuromuscular function by examination and on needle EMG. The MRI scan showed moderate degenerative disk disease and slight disk bulging at the C5-6 level. The worst pain now appears to be triggered by pressure over the base of the neck lateral to the seventh cervical spinous process on the right side.
Issue #3: Is the pain regional (eg, mostly myofascial) or widespread?
The pain associated with FMS may be myofascial or widespread (involving all 4 quadrants of the body and the spine). The widespread pain of FMS may be provoked by light touch (allodynia). Myofascial pain may be the primary problem in a muscle or may be associated with a pain trigger, such as a bulging disk or costochondritis. It may be considered significant when it accounts for at least 50% of the patient's total pain experience at a given visit.
Table 4 shows the common regional triggers that can worsen and contribute significantly to FMS pain.The most common triggers are headaches, including migraines; degenerative disk disease; and IBS. Conversely, a patient may present with chronic regional pain syndrome, such as the above patient, when a diagnosis of myofascial pain syndrome should be considered.
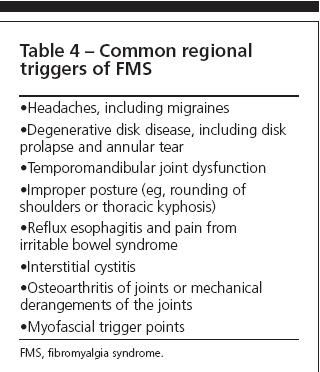
Table 4
Regardless of the presentation, the primary care physician needs to be cognizant of the contribution of a local process to the total pain experience; all factors need to be investigated and managed. Therapy for the local process-physical therapy, bracing, exercise, trigger point injection, acupuncture, and local heat-can have a dramatic effect on the patient's pain level and leads to improvement in the therapeutic response of baseline medications (eg, sleep and pain medications that are used to manage FMS).9,10
Table 5 lists the important differences between myofascial pain syndrome and the allodynia that results from FMS. Attention must be paid in finding not only the regional distribution of myofascial pain but also the appropriate trigger points for therapy and local predisposing factors (eg, regional arthritis, limb length discrepancy, and unusual loading of a skeletal muscle). Functional disturbances (eg, IBS and restless legs syndrome) tend to be less prevalent in patients with regional myofascial pain syndrome.
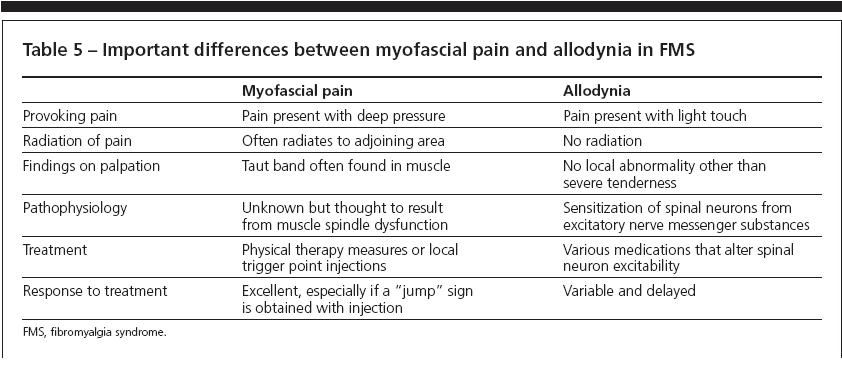
Table 5
Clinical case scenario #2 (continued)
A diagnosis of regional myofascial pain syndrome is made, and trigger point therapy is started. The pain persists even though the patient receives 2 sequential local trigger point injections with 1% lidocaine and 20 mg of triamcinolone.
The patient is referred for physical therapy and acupuncture and receives this therapy once a week. Physical therapy consists of muscle mobilization and strengthening, modalities, and massage 3 times a week.
An overzealous therapist rapidly increases the free weight lifting above the shoulder from a 10-lb weight to a 30-lb weight with 20 to 30 isotonic repetitions. The patient experiences a pain flare-up the day after the first strenuous workout, and he notices that now the pain has crossed over to also involve the left side of the neck.
Issue #4: How to approach exercise?
Exercise is a major component of managing FMS, regional myofascial pain syndrome, and other chronic painful syndromes; there is ample evidence of decreased endurance, fatigue, and muscle pain in FMS with exercise. Most is attributed to muscle deconditioning, but some studies show that anabolic hormones (eg, dihydroepiandrostenedione and testosterone) are reduced in FMS.
Growth hormone (GH) secretion-which occurs predominantly at nighttime during non–rapid eye movement delta wave sleep-is suppressed in FMS, leading to a lack of muscle repair at nighttime and poor muscle endurance and pain during the day. An enhanced somatostatin tone that limits GH release is thought to be present in the brain. Inhibiting the somatostatin tone with exercise and use of pyridostigmine corrects the impaired secretion of GH.
In a double-blind trial of several months' duration, GH administration decreased patients' overall level of pain and tender points on examination.11 However, the effects were short-lived and the cost of the GH medication is prohibitive. Therefore, treatment is directed at improving sleep and encouraging exercise to improve muscle endurance.
The ability to exercise is quite variable in patients with FMS. Some can barely walk a few blocks because of an increase in myalgia that occurs postexercise. Therefore, exercise should be tailored to the specific patient's ability.
Some forms of exercise appear to work better than others in patients with FMS. Evidence-based approaches suggest that aquatherapy holds the most promise.
Clinical case scenario #2 (continued)
The patient's pain spreads and in 2 weeks involves the neck, shoulders, and upper back on both sides, even though he is using analgesic medications (eg, ibuprofen) around the clock and muscle relaxants (eg, cyclobenzaprine) at bedtime. The patient reports disturbed sleep, with an inability to fall asleep because of pain and frequent awakening at night because of pain felt when he lies in 1 position for more than an hour.
Physical examination reveals tender points in the trapezius muscle at the occiput, the base of the neck, and the midpoint of the trapezius between the shoulder and neck. The trigger points radiate pain into the upper arms bilaterally. Myofascial pain is present between the shoulder blades and over the subscapularis muscle in the axillary areas bilaterally. Pain with light touch is present over the upper chest wall at the site of attachment of the pectoral muscles and over both subacromial bursa areas.
The ibuprofen is substituted with tramadol in an extended release formulation to manage the around-the-clock pain.The dose is gradually titrated up to achieve pain levels of 3 to 4 on a 0-to-10 pain scale. Intermittent spiking of pain lasting a few hours continues to occur in the late afternoon, after aquatherapy, and at nighttime. Pain on light touch continues to be present over the chest wall.
The breakthrough pain is managed with immediate-release hydrocodone twice a day before aquatherapy and at bedtime. Cyclobenzaprine is discontinued and pregabalin is started in low doses twice a day and titrated to a total daily dose of 300 mg; there is improvement of the pain on light touch and of sleep. A persistent trigger point in the left trapezius is injected with a combination of 20 mg of methylprednisolone acetate and 1 cc of lidocaine. The patient continues to receive these medications at 6 months and has achieved good pain control, improved sleep function, and return to work full-time.
Issue #5: How to determine medication approaches?
A fundamental approach to treating any patient who has chronic pain is to ascertain whether the pain is around-the-clock or intermittent. The history and physical examination should determine whether the pain is regional or widespread and whether an underlying condition (eg, myelopathy or a metabolic illness) could be the cause. If the pain has been determined to be the result of central sensitization of the dorsal neuron in the spinal cord by the presence of allodynia, widespread trigger points, or more than 11 of the 18 designated tender points for FMS, treatment should be directed at improving sleep architecture and decreasing spinal neuron excitation.12
Approach to improving sleep efficiency and sleep architecture. Sleep efficiency is the ratio of time spent sleeping in bed versus total time spent in bed. Sleep may be improved by decreasing the pain present at the time of sleep onset and using a rapid-acting sleep medication (eg, zolpidem).13 Sleep maintenance may be achieved by combining longer-acting sleep medications (eg, amitriptyline, trazodone, and clonazepam); the need to manage comorbidities, such as migraines and anxiety, should be considered.
Alterations in sleep architecture in FMS include a lack of stage-4 delta wave sleep. Some improvement in delta wave sleep may be obtained by using a medication, such as amitriptyline, tiagabine, or sodium oxybate. A sleep study may be useful in detecting restless legs syndrome, sleep-associated myoclonic jerks, or obstructive sleep apnea, which may be managed with pramipexole, clonazepam, or continuous positive airway pressure, respectively. Daytime naps may not provide sufficient stage-4 sleep; advise patients to make every effort to avoid sleeping during the day.
Decreasing spinal neuron activation. A variety of approaches are used to decrease pain that results from central sensitization of neurons. They can be divided broadly into nonpharmacological and pharmacological methods.
Nonpharmacological methods include the use of appropriate exercise to improve endurance, muscle strength, and endorphin levels in the brain.14 Acupuncture may be used to manage localized myofascial pain, and cognitive-behavioral methods may alter thought processes that are negative and unconducive to proper healing. Physicians need to motivate patients to strive to get better so that they can once again participate in and enjoy their favorite activities.
Medications may be used to target various pathways that have been identified as important in reducing sensitization of neurons (Table 6). They may improve the function of descending inhibitory tracts on spinal neurons (eg, duloxetine, venlafaxine, and tizanidine) or decrease the activity and wind-up of pain-sensing neurons (eg, pregabalin, dextromethorphan, and ketamine).
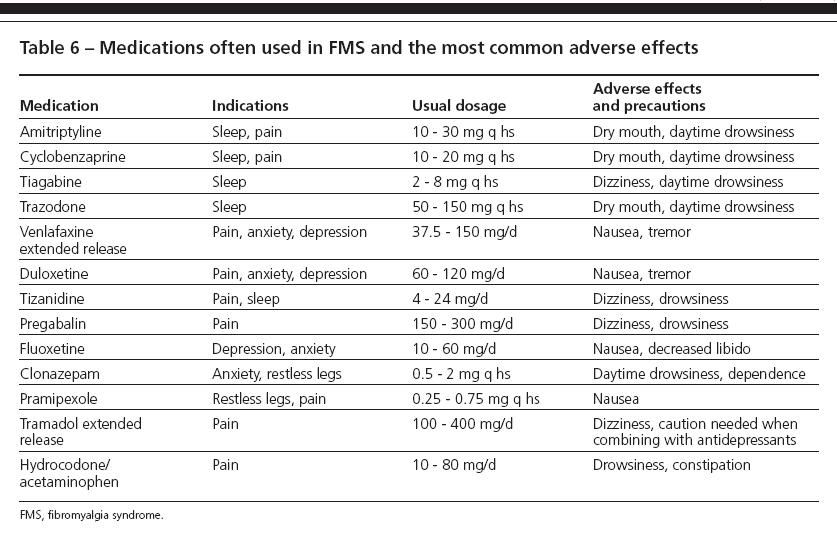
Table 6
Adjunctive medications (eg, SSRIs and benzodiazepines) also may help decrease spinal nerve sensitization; they improve coping mechanisms by decreasing anxiety and depression. Decreases in overall levels of pain in FMS may be achieved by targeting pain triggers-managing a bulging disk with an epidural corticosteroid injection, reducing knee osteoarthritis with local hyaluronic acid injections, preventing migraines with topiramate, and relieving a nagging trigger point with a local corticosteroid/lidocaine combination injection.
Use of long-acting medications to control around-the-clock pain is essential. The least potent yet effective narcotic medication (eg, tramadol extended-release or oxycodone sustained-release preparations) may be needed to control pain that persists in spite of all other attempts to address it. Short-acting pain medications must be used to control breakthrough pain.
References:
References
- Bieber C, Müller KG, Blumenstiel K, et al. A shared decision-making communication training program for physicians treating fibromyalgia patients: effects of a randomized controlled trial. J Psychosom Res. 2008;64:13-20.
- MacFarlane GJ, Thomas E, Papageorgiou AC, et al. The natural history of chronic pain in the community: a better prognosis than in the clinic? J Rheumatol. 1996;23:1617-1620.
- Yunus MB. Fibromyalgia syndrome: clinical features and spectrum. J Musculoskel Pain. 1994;2:5-21.
- Wolfe F, Ross K, Anderson J, et al. The prevalence and characteristics of fibromyalgia in the general population. Arthritis Rheum. 1995;38:19-28.
- Wallace DJ, Clauw DJ, eds. Fibromyalgia and Other Central Pain Systems. Philadelphia: Lippincott Williams & Wilkins; 2005.
- Wallace DJ, Clauw DJ, Hallegua DS. Addressing behavioral abnormalities in fibromyalgia. J Musculoskel Med. 2005;22:562-579.
- Aaron LA, Bradley LA, Alarcon GS, et al. Psychiatric diagnoses in patients with fibromyalgia are related to health care-seeking behavior rather than to illness. Arthritis Rheum. 1996;39:436-445.
- Wallace DJ, Gotto J. Hypothesis: bipolar illness with complaints of chronic musculoskeletal pain is a form of pseudofibromyalgia. Semin Arthritis Rheum. 2008;37:256-259.
- Travell JG, Simons DG. Myofascial Pain and Dysfunction: The Trigger Point Manual. Vol 1: The Upper Extremities. Baltimore: Lippincott Williams & Wilkins; 1983.
- Ready LB, Kozody R, Barsa JE, Murphy TM. Trigger point injections vs jet injection in the treatment of myofascial pain. Pain. 1983;15:201-206.
- Jones KD, Deodhar P, Lorentzen A, et al. Growth hormone perturbations in fibromyalgia: a review. Semin Arthritis Rheum. 2007;36:357-379.
Hallegua DS, Wallace DJ. Comprehensive management of fibromyalgia.
J Musculoskel Med.
2005;22:382-391.
- Burckhardt CS. Nonpharmacologic management strategies in fibromyalgia. Rheum Dis Clin North Am. 2002;28:291-304.
- Barkhuizen A. Rational and targeted pharmacologic treatment of fibromyalgia. Rheum Dis Clin North Am. 2002;28:261-290.