Low back pain management: Approaches to treatment
anagement of low back pain (LBP) includes a variety of approaches. The clinician should involve patients in the discussion and encourage them to become active participants. Bed rest has many deleterious effects; remaining active is more effective for patients with acute or subacute LBP.
Management of low back pain (LBP) involves a multifaceted approach with the goals of relieving the patient’s pain and restoring normal function. With a detailed evaluation, clinicians may establish an appropriate diagnosis and formulate a targeted treatment plan. Approaches to treatment include lifestyle modification, medications, physical therapy and various modalities, psychological counseling and, where appropriate, interventional procedures.
In this 2-part article, we provide an update on approaches to LBP management. The first part (“Low back pain management: Making the diagnosis,” The Journal of Musculoskeletal Medicine, July 2010, page 249) focused on patient evaluation. In this second part, we explore the treatment options.
LIFESTYLE MODIFICATION
Education
After a thorough history, physical examination, and review of diagnostic testing, the clinician should involve the patient in the discussion of the treatment plan. This is the appropriate time to review the pertinent anatomy, biomechanics, and underlying pain generators of the spine.
Discussion of the treatment plan should include a description and rationale for additional diagnostic testing, if necessary, and medications, therapeutic exercises, or interventional procedures (Figure).
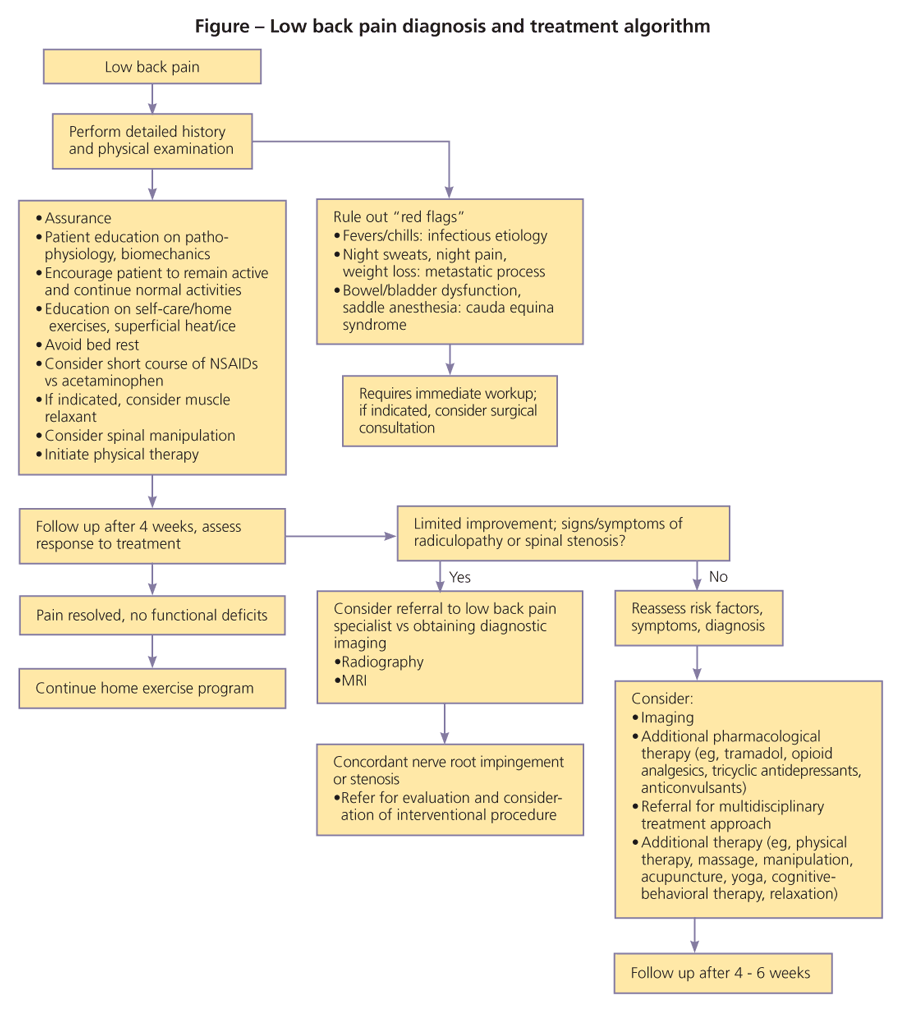
The patient should be encouraged to become an active participant in his or her treatment. The clinician can engage the patient in the discussion with a review of proper posture, spine biomechanics in activities of daily living, and simple methods to minimize symptoms.
Bed rest
Historically, bed rest was the treatment for patients with LBP. Although some benefit may be gained from reducing intradiskal pressure while the patient is prone, bed rest has many deleterious effects on bone, connective tissue, muscle, and cardiovascular fitness. The proactive approach emphasizes activity modification rather than strictly bed rest.
Remaining active is more effective than bed rest for patients who have acute or subacute LBP.1,2 The patient should be instructed to avoid activities that increase intradiskal pressure, such as sitting, bending, and lifting.
TRACTION
Lumbar traction has long been a preferred method for managing lumbar disk problems. About 1.5 times a patient's body weight is required to develop distraction of the vertebral bodies. In a recent literature review, there was no evidence to support lumbar traction as a treatment.3 In fact, there was the suggestion that sustained traction might cause more harm than good. However, a randomized controlled trial conducted by Fritz and associates4 showed that there may be a subgroup of patients who may benefit from traction in the short term. This subgroup is characterized by the presence of leg symptoms, signs of nerve root compression, and either peripheralization with extension movements or a positive crossed straight-leg raising test result.
LUMBAR BRACING
Braces have been used prophylactically to prevent injury to the lumbosacral spine, and they purportedly help manage existing pathology. The use of lumbar bracing has not demonstrated efficacy as a means to prevent LBP in the workplace.5
Lumbar supports are not more effective than other marginal therapies in reducing LBP, and there is minimal evidence to support their use.6 Lumbar bracing may help prevent reinjury by serving as a kinesthetic reminder for using proper biomechanics when lifting or bending, although this has not been demonstrated in the literature.
EXERCISE
There is no good scientific evidence to support therapeutic exercise in acute LBP. In subacute back pain, however, an intensive interdisciplinary rehabilitation program that includes physician consultation with psychological, physical therapy, and social/vocational intervention has been shown to be moderately effective.7 In patients with chronic LBP, programs that incorporate tailoring to individual needs, supervision, stretching, and strengthening are associated with the best outcomes.8
The overall goals of exercise programs for LBP are to reduce pain, restore normal motion, and develop muscular strength of the trunk and spine sufficient to diminish stress to the intervertebral disk and static stabilizers of the spine.9 In addition, therapy should be directed to the patient who demonstrates reasonable understanding of his pain and good technique in performing the exercises on his own and to implementation of an appropriate home exercise program.
MANIPULATION
There is conflicting evidence about spinal manipulation and manual mobilization in the management of LBP, although recent clinical guidelines suggest that spinal manipulation in the hands of trained professionals provides a small to moderate short-term benefit in relieving pain.10 Moderate evidence suggests that the effect of manipulation in combination with strengthening exercise is similar to the effect of prescription NSAIDs with exercise in both the short and long terms.11
Spinal manipulation has not been found to be more effective than other treatments, such as analgesics, exercise, and physical therapy. However, there appears to be a subgroup of patients with LBP for whom spinal manipulation results in significant reductions in pain and disability.12-14 Manipulation should be performed in conjunction with and to facilitate an active physical therapy program.
MEDICATIONS
Patients with LBP have been treated with medications in a number of classes, including acetaminophen, NSAIDs, muscle relaxants, opioid analgesics, and oral corticosteroids. Each agent has unique trade-offs between risk and anticipated benefits. Therefore, before prescribing a medication, the clinician should be aware of its contraindications, common adverse effects, and mechanism of action.
Acetaminophen
This para-aminophen derivative has analgesic and antipyretic effects equal to those of aspirin, but it has weak anti-inflammatory effects. Acetaminophen is relatively inexpensive and is available without a prescription. It is effective for mild to moderate pain but lacks other desirable effects on inflammation and muscle spasm. In acute LBP, studies have shown no difference between acetaminophen and no treatment,15 and no clear difference was seen in pain relief between acetaminophen and NSAIDs.16,17
Acetaminophen generally is not a first-line medication for LBP unless there are contraindications to other medications. Prolonged use is contraindicated because of the potential for liver toxicity.
NSAIDs
These agents are a reasonable first-line treatment for acute LBP because of their combined analgesic and anti-inflammatory effects. Nonselective NSAIDs have been shown to be more effective for pain relief than acetaminophen.17 The dose needed to produce anti-inflammatory effects differs substantially from that for analgesic effects. These medications often are taken intermittently, and a significant level is not sustained to take advantage of the anti-inflammatory properties. Patients should be advised to take the medications at regular intervals to maximize both the analgesic and the anti-inflammatory effects.
No one NSAID has been shown to be more effective than the others in terms of pain relief for patients with LBP. In a systematic review, the selective cyclooxygenase-2 inhibitors showed fewer adverse effects than the traditional NSAIDs.18 The major adverse reactions to NSAIDs include GI bleeding and renovascular damage. NSAIDs also have been shown to slow bone and tissue healing.19 Prolonged use should be avoided to minimize the associated risks. The lowest effective dose is recommended for the shortest duration that is necessary.20
Antidepressants
There is good evidence to support the analgesic effect of tricyclic antidepressants in the treatment of patients with LBP. In a systematic review, tricyclic antidepressants were more effective than placebo in controlling pain.21 Other antidepressants in the selective serotonin reuptake inhibitor class and trazodone have been shown to be no more effective than placebo.22,23
Antidepressants typically take up to 4 weeks to achieve effect, but whether the same time is required to achieve pain relief is unknown.23 Of note, depression is a common problem in patients with chronic LBP, and it should be addressed appropriately.24
Muscle relaxants
These often are prescribed in the management of acute LBP to relieve pain, improve range of motion, and interrupt the pain-spasm-pain cycle. They have been shown to be more effective when they are used in conjunction with NSAIDs.25
Tizanidine has been shown to be effective in the management of acute LBP.26 Other studies have not determined superiority for any particular muscle relaxant in terms of benefit or adverse effects. The most frequently reported effect of muscle relaxants is sedation; they usually are prescribed at bedtime to take advantage of this property.
Opioid analgesics
The use of opioid analgesics in the management of LBP should be limited to pain that is unresponsive to other treatments or when other medications are contraindicated. A careful risk-benefit analysis should be considered before starting these medications because of the potential risk of aberrant drug-related behaviors with long-term use in patients who have a history of or predilection to addiction or abuse. There is no evidence to support the use of one opioid versus another.27 Prolonged or repeated use of opioids is not necessary for most patients who have acute LBP.
Tramadol
This centrally acting analgesic has a combined mechanism of binding to µ receptors and a mixed serotonin/norepinephrine reuptake inhibitor. Although no studies have compared tramadol with acetaminophen, NSAIDs, or opioid analgesic monotherapy, there is evidence to suggest that tramadol is effective for short-term pain relief and improved function.28
Antiseizure medications
Gabapentin, an antiepileptic medication widely used in the management of neuropathic pain, has been shown to provide short-term benefit in patients with radicular pain.29,30 The most frequently noted adverse effects include drowsiness, loss of energy, and dizziness. There is insufficient evidence for or against other antiepileptic medications in the management of LBP with or without radiculopathy.20
Oral corticosteroids
The use of oral corticosteroids in the management of LBP is unsupported in the literature, especially when there are radicular symptoms, because these agents have not been shown to be more effective than placebo.20,31-34 In a double-blind placebo-controlled trial, Holve and Barkan35 demonstrated that patients treated with a tapering course of prednisone have a more rapid return to baseline in pain, mental well-being, and disability scores and require fewer subsequent epidural injections. There was no difference in physical examination findings, the use of NSAIDs or other pain medications, or return to work. Better studies clearly are needed to determine the role and effectiveness of oral corticosteroids in LBP, particularly for lumbar radiculopathy.
MODALITIES
Modalities offer an adjunct to evidence-based treatment in patients with LBP. However, the evidence to support their use is generally poor, despite their widespread use.
Transcutaneous electrical nerve stimulation (TENS)
TENS, the use of electrical impulses over surface electrodes to provide symptomatic relief by modifying pain perception, has not been shown to be effective for chronic LBP.20 In recently published American Academy of Neurology guidelines, TENS is not recommended for chronic LBP because it has not been shown to be more effective than placebo.36
Electrical stimulation
High-voltage pulsed galvanic stimulation has been used to reduce muscle spasm and edema in patients with acute LBP. The use of electrical stimulation typically is limited to the initial stages of treatment to allow patients to progress to more active treatments in restoring normal range of motion and strength. There is insufficient scientific evidence for or against its use in patients who have LBP.37
Ultrasonography
This deep-heating modality has been shown to improve the distensibilty of connective tissue and facilitate stretching.38 Ultrasonography is contraindicated in acute inflammatory conditions because it may exacerbate the inflammatory response. In addition, this modality is contraindicated over a previous laminectomy site.
There is insufficient evidence for or against the use of ultrasonography in LBP.37 It may be best used to improve segmental limitations in range of motion by facilitating soft tissue mobilization and stretching with a skilled physical therapist.25
Superficial heat
This modality produces effects to a depth of 1 to 2 cm and has been shown to reduce muscle spasm and pain. Heat packs often are used in combination with electrical stimulation therapy. Heat wrap therapy has been shown to be more effective than placebo in short-term pain relief and functional status.39 These effects typically last for less than 1 week and are not more effective than exercise.
THERAPEUTIC INJECTIONS
Trigger point injections
Myofascial trigger points are thought to be hyperirritable foci within muscles and fasciae that are associated with taut muscle bands. Trigger points are identified on palpation, which produces a focal twitch response and referred pain distal to the site of muscle irritability. Palpation examinations have poor inter-rater reliability.40 In an assessment of intra-rater reliability, local twitch response and referred pain varied from one session to the next; taut bands, tender points, and ‘‘jump sign'' remained consistent.41
Trigger points are managed initially with stretching; restoration of normal posture and biomechanics; and modalities, such as superficial heat or cryotherapy. Muscle relaxants also may be useful.
Trigger point injections may be considered when more conservative measures have not succeeded. However, there is no evidence that injections of normal saline with corticosteroids are more effective than normal saline alone. The benefit may result from needling alone or from placebo or nonspecific effects.
In one study, there was no difference among dry needling, injection with lidocaine, lidocaine with corticosteroid, and vasocoolant spray with acupressure.42,43 As a result, the type of injections, appropriate dosage, and interval are not known.
Trigger point injections remain an option for myofascial-related LBP resistant to conservative treatments. They should be limited and used in conjunction with an appropriate functional rehabilitation program.
Epidural corticosteroid
injections
These injections are a frequently performed interventional procedure aimed at reducing pain and inflammation resulting from disk herniation and subsequent nerve root irritation. The effectiveness of these injections is increased if they are used in the first weeks after the onset of pain and followed with an active exercise program. Even though they are used widely, good scientific evidence for their use in the management of LBP is limited. In a multicenter randomized controlled trial, epidural corticosteroid injections offered transient benefit in symptoms at 3 weeks in patients with sciatica but no sustained benefits in terms of pain, function, or the need for surgery.44
Epidural injections should be used when clinical evidence based on the history and physical examination correlates well with radiographic evidence. To minimize the risks, they should be performed with fluoroscopic guidance and contrast enhancement. There is no indication for performing injections in “series.” Typically, 1 or 2 injections are sufficient to improve the radicular pain symptoms and facilitate a successful course of rehabilitation.
Facet injections
Although lumbar facet joints are a potential source of LBP, there is a poor correlation between history and physical examination findings and true facet-mediated LBP. In addition, diagnostic imaging is unreliable for identifying underlying facet joint pathology. In spite of various attempts reported in the literature, an identifying clinical feature of facet-mediated pain has yet to be found. Thus, the only means of diagnosis is through facet joint blocks.
Facet joint injections with corticosteroids have not been shown to be more effective than placebo in controlling pain.45,46 Therefore, lumbar medial branch blocks or radiofrequency neurotomies remain a potential treatment, given an appropriate patient response to diagnostic blockade.46
Sacroiliac joint injections
The sacroiliac joint is a potential pain generator in the lumbar spine, with an overlapping referral pain pattern around the posterosuperior iliac spine. Sacroiliac joint–mediated pain should be considered in patients for whom a comprehensive rehabilitation program and a trial of NSAIDs, ice/heat, and mobilization or manipulation have not succeeded. Injection should be considered for both diagnostic and therapeutic purposes; if the results are positive, it should be followed with an active physical therapy program.
Prolotherapy
This therapy involves injection of solution to promote healing of loose tissue, ligaments, tendons, and joint capsules. Prolotherapy remains poorly studied and validated in spite of a long history of use to manage various conditions thought to be the result of ligamentous instability.
In the management of LBP, prolotherapy has been shown to be effective in combination with spinal manipulation therapy.47,48 It has yet to be studied as a single treatment without co-interventions.
ACUPUNCTURE
This modality has been used to manage various conditions for more than 2000 years. Evidence for the use of acupuncture in the management of acute LBP is sparse,49 but it has been shown to be effective in the management of chronic LBP.50,51 The literature on acupuncture is not of high quality.49
PSYCHOLOGICAL
COUNSELING
Cognitive-behavioral therapy is an effective component in the treatment of patients who have chronic pain.52 However, it needs to be combined with other therapeutic components, such as physical therapy, to deal with physical deconditioning issues. Currently, there are no studies that directly address what combination of components might provide the best therapeutic outcomes for what type of chronic pain syndrome.52
SPINAL CORD STIMULATION
With this modality, an implantable device is used primarily to manage failed back surgery syndrome, complex regional pain syndrome, and chronic back pain. As the number of low back surgeries increases, so do the number and the use of spinal cord stimulation therapies.53
In a recent systematic review, Frey and associates53 found strong evidence for the clinical use of spinal cord stimulation in failed back surgery syndrome in terms of pain relief and cost-effectiveness. Spinal cord stimulation does involve risk, which is estimated to exist in up to 43% of patients. The most common complications include electrode or lead problems, infection, generator problems, extension cable problems, and cerebrospinal fluid leakage.53
CONCLUSIONS
LBP is a frequently encountered complaint associated with great costs that continue to increase. Most patients improve with time if they remain active. Physicians need to have a clear understanding of the pertinent anatomy and physiology of the spine and correlate this with their findings in the history and on physical examination to develop a good differential diagnosis and a proper treatment plan. The literature may help guide the decision-making process, but much of the evidence is not well validated. The best evidence is for a short course of NSAIDs, active exercise and, as soon as possible, a return to normal activities. Physical therapy, injections, and other approaches to treatment may help in properly selected patients.
References:
References
1. Hagen KB, Hilde G, Jamtvedt G, Winnem M. Bed rest for acute low back pain and sciatica. Cochrane Database Sys Rev. 2004;(4):CD001254. Article withdrawn June 16, 2010.
2. Hilde G, Hagen KB, Jamtvedt G, Winnem M. WITHDRAWN: advice to stay active as a single treatment for low-back pain and sciatica. Cochrane Database Syst Rev.2007;(2):CD003632.
3. Gay RE, Brault JS. Evidence-informed management of chronic low back pain with traction therapy. Spine J. 2008;8:234-242.
4. Fritz JM, Delitto A, Erhard RE. Comparison of classification-based physical therapy with therapy based on clinical practice guidelines for patients with acute low back pain: a randomized clinical trial. Spine (Phila Pa 1976). 2003;28:1363-1372.
5. Woodhouse ML, McCoy RW, Redondo DR, Shall LM. Effects of back support on intra-abdominal pressure and lumbar kinetics during heavy lifting. Hum Factors. 1995;37:582-590.
6. Jellema P, van Tulder MW, van Poppel MN, et al. Lumbar supports for prevention and treatment of low back pain: a systematic review within the framework of the Cochrane Back Review Group. Spine (Phila Pa 1976). 2001;26:377-386.
7. Karjalainen K, Malmivaara A, van Tulder M, et al. Multidisciplinary biopsychosocial rehabilitation for subacute low back pain in working-age adults: a systematic review within the framework of the Cochrane Collaboration Back Review Group. Spine (Phila Pa 1976). 2001;26:262-269.
8. Hayden JA, van Tulder MW, Tomlinson G. Systematic review: strategies for using exercise therapy to improve outcomes in chronic low back pain. Ann Intern Med. 2005;142:776-785.
9. Saal JA. Dynamic muscular stabilization in the nonoperative treatment of lumbar pain syndromes. Orthop Rev. 1990;19:691-700.
10. Assendelft WJ, Morton SC, Yu EI, et al. Spinal manipulative therapy for low back pain: a meta-analysis of effectiveness relative to other therapies. Ann Intern Med. 2003;138:871-881.
11. Bronfort G, Haas M, Evans R, et al. Evidence-informed management of chronic low back pain with spinal manipulation and mobilization. Spine J. 2008;8:213-225.
12. Flynn T, Fritz J, Whitman J, et al. A clinical prediction rule for classifying patients with low back pain who demonstrate short-term improvement with spinal manipulation. Spine (Phila Pa 1976). 2002;27:2835-2843.
13. Childs JD, Fritz JM, Flynn TW, et al. A clinical prediction rule to identify patients with low back pain most likely to benefit from spinal manipulation: a validation study. Ann Intern Med. 2004;141:920-928.
14. Cleland JA, Fritz JM, Kulig K, et al. Comparison of the effectiveness of three manual physical therapy techniques in a subgroup of patients with low back pain who satisfy a clinical prediction rule: a randomized clinical trial. Spine (Phila Pa 1976). 2009;34:2720-2729.
15. Milgrom C, Finestone A, Lev B, et al. Overexertional lumbar and thoracic back pain among recruits: a prospective study of risk factors and treatment regimens. J Spinal Disord. 1993;6:187-193.
16. van Tulder MW, Scholten RJ, Koes BW, Deyo RA. Non-steroidal anti-inflammatory drugs for low back pain [published updates appear in Cochrane Database Syst Rev. 2008;(1):CD000396 and 2006;(2):CD000396]. Cochrane Database Syst Rev. 2000;(2):CD000396.
17. van Tulder MW, Scholten RJ, Koes BW, Deyo RA. Nonsteroidal anti-inflammatory drugs for low back pain: a systematic review within the framework of the Cochrane Collaboration Back Review Group. Spine (Phila Pa 1976). 2000;25:2501-2513.
18. Roelofs PD, Deyo RA, Koes BW, et al. Non-steroidal anti-inflammatory drugs for low back pain: an updated Cochrane review. Spine (Phila Pa 1976). 2008;33:1766-1774.
19. Riew KD, Long J, Rhee J, et al. Time-dependent inhibitory effects of indomethacin on spinal fusion. J Bone Joint Surg. 2003;85A:632-634.
20. Chou R, Qaseem A, Snow V, et al; Clinical Efficacy Assessment Subcommittee of the American College of Physicians; American College of Physicians; American Pain Society Low Back Pain Guidelines Panel. Diagnosis and treatment of low back pain: a joint clinical practice guideline from the American College of Physicians and the American Pain Society [published correction appears in Ann Intern Med. 2008;148:247-248]. Ann Intern Med. 2007;147:478-491.
21. Staiger TO, Gaster B, Sullivan MD, Deyo RA. Systematic review of antidepressants in the treatment of chronic low back pain. Spine (Phila Pa 1976). 2003;28:2540-2545.
22. Salerno SM, Browning R, Jackson JL. The effect of antidepressant treatment on chronic back pain: a meta-analysis. Arch Intern Med. 2002;162:19-24.
23. Malanga GA, Dennis RL. Use of medications in the treatment of acute low back pain. Clin Occup Environ Med. 2006;5:643-653, vii.
24. Bair MJ, Robinson RL, Katon W, Kroenke K. Depression and pain comorbidity: a literature review. Arch Intern Med. 2003;163:2433-2445.
25. Malanga GA, Nadler SF. Nonoperative treatment of low back pain. Mayo Clin Proc. 1999;74:1135-1148.
26. van Tulder MW, Touray T, Furlan AD, et al; Cochrane Back Review Group. Muscle relaxants for nonspecific low back pain: a systematic review within the framework of the cochrane collaboration. Spine (Phila Pa 1976). 2003;28:1978-1992.
27. Chou R, Clark E, Helfand M. Comparative efficacy and safety of long-acting oral opioids for chronic non-cancer pain: a systematic review. J Pain Symptom Manage. 2003;26:1026-1048.
28. Schnitzer TJ, Gray WL, Paster RZ, Kamin M. Efficacy of tramadol in treatment of chronic low back pain. J Rheumatol. 2000;27:772-778.
29. McCleane GJ. Does gabapentin have an analgesic effect on background, movement and referred pain? A randomised, double-blind, placebo controlled study. Pain Clinic. 2001;13:103-107.
30. Yildirim K, Sisecioglu M, Karatay S, et al. The effectiveness of gabapentin in patients with chronic radiculopathy. Pain Clinic. 2003;15:213-218.
31. Finckh A, Zufferey P, Schurch MA, et al. Short-term efficacy of intravenous pulse glucocorticoids in acute discogenic sciatica: a randomized controlled trial. Spine (Phila Pa 1976). 2006;31:377-381.
32. Friedman BW, Holden L, Esses D, et al. Parenteral corticosteroids for Emergency Department patients with non-radicular low back pain. J Emerg Med. 2006;31:365-370.
33. Haimovic IC, Beresford HR. Dexamethasone is not superior to placebo for treating lumbosacral radicular pain. Neurology. 1986;36:1593-1594.
34. Porsman O, Friis H. Prolapsed lumbar disc treated with intramuscularly administered dexamethasonephosphate: a prospectively planned, double-blind, controlled clinical trial in 52 patients. Scand J Rheumatol. 1979;8:142-144.
35. Holve RL, Barkan H. Oral steroids in initial treatment of acute sciatica. J Am Board Fam Med. 2008;21:469-474.
36. Dubinsky RM, Miyasaki J. Assessment: efficacy of transcutaneous electric nerve stimulation in the treatment of pain in neurologic disorders (an evidence-based review): report of the Therapeutics and Technology Assessment Subcommittee of the American Academy of Neurology. Neurology. 2010;74:173-176.
37. Poitras S, Brosseau L. Evidence-informed management of chronic low back pain with transcutaneous electrical nerve stimulation, interferential current, electrical muscle stimulation, ultrasound, and thermotherapy. Spine J. 2008;8:226-233.
38. Lehmann JF, Masock AJ, Warren CG, Koblanski JN. Effect of therapeutic temperatures on tissue extensibility. Arch Phys Med Rehabil. 1970;51:481-487.
39. French SD, Cameron M, Walker BF, et al. Superficial heat or cold for low back pain. Cochrane Database Syst Rev. 2006;(1):CD004750.
40. Nice DA, Riddle DL, Lamb RL, et al. Intertester reliability of judgments of the presence of trigger points in patients with low back pain. Arch Phys Med Rehabil. 1992;73:893-898.
41. Al-Shenqiti AM, Oldham JA. Test-retest reliability of myofascial trigger point detection in patients with rotator cuff tendonitis. Clin Rehabil. 2005;19:482-487.
42. Malanga G, Wolff E. Evidence-informed management of chronic low back pain with trigger point injections. Spine J. 2008;8:243-252.
43. Garvey TA, Marks MR, Wiesel SW. A prospective, randomized, double-blind evaluation of trigger-point injection therapy for low back pain. Spine (Phila Pa 1976). 1989;14:962-964.
44. Arden NK, Price C, Reading I, et al; WEST Study Group. A multicentre randomized controlled trial of epidural corticosteroid injections for sciatica: the WEST study. Rheumatology (Oxford). 2005;44:1399-1406.
45. Ogsbury JS 3rd, Simon RH, Lehman RA. Facet “denervation”in the treatment of low back syndrome. Pain. 1977;3:257-263.
46. Bogduk N. Evidence-informed management of chronic low back pain with facet injections and radiofrequency neurotomy. Spine J. 2008;8:56-64.
47. Ongley MJ, Klein RG, Dorman TA, et al. A new approach to the treatment of chronic low back pain. Lancet. 1987;2:143-146.
48. Klein RG, Eek BC, DeLong WB, Mooney V. A randomized doubleblind trial of dextrose-glycerin-phenol injections for chronic, low back pain. J Spinal Disord. 1993;6:23-33.
49. Ammendolia C, Furlan AD, Imamura M, et al. Evidence-informed management of chronic low back pain with needle acupuncture. Spine J. 2008;8:160-172.
50. Furlan AD, van Tulder M, Cherkin D, et al. Acupuncture and dry-needling for low back pain: an updated systematic review within the framework of the cochrane collaboration. Spine (Phila Pa 1976). 2005;30:944-963.
51. Manheimer E, White A, Berman B, et al. Meta-analysis: acupuncture for low back pain. Ann Intern Med. 2005;142:651-663.
52. Gatchel RJ, Rollings KH. Evidence-informed management of chronic low back pain with cognitive behavioral therapy. Spine J. 2008;8:40-44.
53. Frey ME, Manchikanti L, Benyamin RM, et al. Spinal cord stimulation for patients with failed back surgery syndrome: a systematic review. Pain Physician. 2009;12:379-397.