Achilles tendon evaluation and repair
ABSTRACT: Achilles tendon injuries are increasing as the population ages yet remains physically active later in life. Early diagnosis and management are important for good long-term outcome, but many injuries are missed or misdiagnosed on initial evaluation.
Injury to the Achilles tendon, the thickest and strongest tendon in the human body, is increasing in frequency. Acute rupture of the Achilles tendon, the most common acute injury, has been reported to affect about 18 persons per 100,000 each year.1 As the population ages and more persons remain active later in life, the rate of tendinous injuries is increasing and probably will continue to do so.
Typically, patients with Achilles tendon injury are 30- to 40-year-old men who present with a complaint of sudden ankle or calf pain that they experienced during recreational sports activity. Basketball, tennis, and football are the sports in which Achilles tendon injuries occur most frequently; 50% to 75% of acute injuries may be attributed to sports activity. The tendon may be disrupted at its musculotendinous origin, at the insertion on the calcaneus or, most commonly, within the substance of the tendon itself.
A thorough understanding of the risk factors, presentation, and treatment of injury to the Achilles tendon should allow prompt recognition of the injury and referral to an orthopedic surgeon for treatment. Given the relative lack of symptom severity associated with acute rupture of the Achilles tendon, however, many of these injuries are missed on initial evaluation.2 With an accurate history and thorough physical examination, the injury should not escape detection.
Early management of Achilles tendon injuries is important for good long-term outcome. There is a relatively large body of literature but no consensus about the optimal management approach. The standard of care for acute rupture of the tendon is evolving-more and more surgeons prefer operative intervention to minimize rerupture and increase the probability of the patient’s return to sports activity. Patients may seek an operative option to decrease the recovery process or simply for convenience.
Correct identification of Achilles tendon injuries by primary care physicians is a vital first step in effective treatment of this common problem. In this article, we highlight key concepts that help make the diagnosis. We also offer a brief but detailed description of the various methods for management of acute Achilles rupture that should help primary care physicians inform and counsel their patients before referral.
ANATOMY
The Achilles tendon is formed from a confluence of the gastrocnemius and soleus muscles (Figure). The soleus, the more anterior of the muscles, has its origin on the tibia; the gastrocnemius originates from the femur and, therefore, crosses the knee, ankle, and subtalar joints. Because the fibers have about a 90° lateral twist as they travel distally, the gastrocnemius component of the tendon tends to insert laterally; the medial attachment is derived predominantly from the soleus. The contributions of each muscle remain relatively distinct for the length of the tendon.
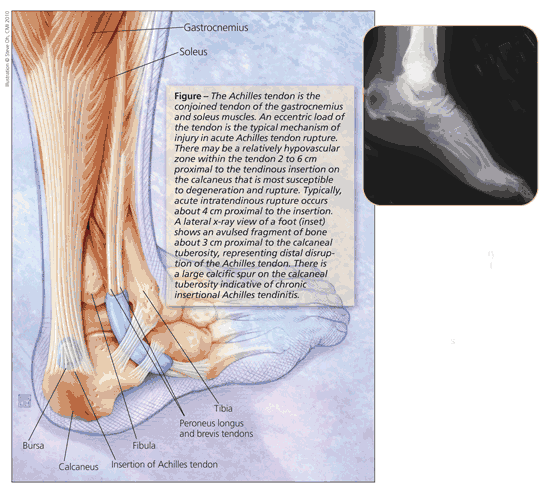
Type 1 collagen accounts for about 65% to 80% of the tendon’s dry mass. The epitenon, a thin membrane of connective tissue, surrounds the tendon and provides its vascular, lymphatic, and nervous connections. Surrounding the epitenon is another connective tissue layer, the paratenon. Fluid within this layer allows for more efficient gliding of the tendon.
Dye injection studies have shown the presence of a relatively hypovascular zone within the Achilles tendon 2 to 6 cm proximal to the tendinous insertion on the calcaneus.3 Some observers maintain that this “watershed” region of the tendon is most susceptible to degeneration and acute rupture. However, subsequent studies that used laser color Doppler flowmetry have shown a more even distribution of blood flow throughout the tendon and called this theory into question.4
ACUTE INTRATENDINOUS RUPTURE
Cause of injury
The typical mechanism of injury in acute Achilles tendon rupture is an eccentric load of the tendon. Essentially, the tendon is pulled in opposite directions: the contracting gastrocsoleus complex attempts to plantar flex the foot while external forces dorsiflex the foot (eg, when a basketball player tries to jump suddenly and forcefully while landing from a previous leap).
Arner and Lindholm5 reviewed the mechanism of injury for 92 acute ruptures and found 3 main causes. First, 53% of the ruptures occurred when the weight-bearing forefoot was pushing off while the knee was extending. This is common in sports that require rapid stops and starts, quick jumps, or sprint starts (eg, tennis, basketball, and football). A second mechanism (17% of the ruptures) was sudden, unexpected dorsiflexion of the ankle (eg, when a person falls off a ladder or steps into a hole). The third category, violent dorsiflexion of the foot (eg, when a person falls from a height), accounted for 10% of the acute ruptures.
The vast majority of persons with a ruptured Achilles tendon do not have antecedent pain in the tendon. However, histological studies have indicated that acute rupture is more probable in an otherwise abnormal tendon.6 Kannus and Jzsa7 found that 865 of 891 ruptured tendons (97%) showed signs of degeneration, compared with 149 of 445 control tendons (33%).
A 200-fold increase in risk of tearing of the contralateral tendon in persons who previously ruptured their Achilles tendon also lends evidence to the theory that certain characteristics predispose a tendon to injury. Several case reports describe a risk of tendon rupture associated with systemic use of fluoroquinolone antibiotics or local injection of corticosteroid into the tendon. The peak incidence of acute rupture, seen in middle-aged men who participate in recreational sports rather than in older men, suggests that there is also a mechanical cause of injury. Anecdotally, we are seeing more women in this same age category with acute Achilles ruptures, likely a result of women engaging in more high-demand activities than in previous years.
Clinical presentation
In the history, patients often describe having felt a “pop” in the area of the Achilles tendon. Other patients report the sensation of being hit or kicked in the back of the ankle, followed by weakness, pain, and apprehension with plantar flexion.
Typically, the patient cannot return to play. However, most patients retain the ability to walk with a slight limp and do not experience significant pain. As a result, many patients self-diagnose a “sprain” and wait to seek medical evaluation.
Up to 25% of acute Achilles ruptures are misdiagnosed on initial evaluation.2 Usually, injured patients are thought to have an ankle sprain. The differential diagnosis includes problems that may cause acute calf or ankle pain, such as partial Achilles tendon rupture, deep venous thrombosis, thrombophlebitis, ankle arthritis, and Achilles tendinitis. An awareness of the demographics of injury coupled with an accurate history should raise the index of suspicion for an acute rupture of the Achilles tendon and guide the clinician to a focused physical examination for confirmation of the diagnosis.
Physical examination
On examination, the clinician will note some degree of swelling, ecchymosis, and tenderness overlying the tendon. However, this can be quite variable. Often, there is a palpable defect, or “soft spot,” within the substance of the tendon that typically is located 4 to 6 cm proximal to the insertion of the Achilles tendon on the heel.
The presence of weak, active ankle plantar flexion should not lead the examiner to rule out complete rupture. In fact, active ankle plantar flexion usually is preserved, because the patient may use the tibialis posterior, peroneal, flexor digitorum, or flexor hallucis longus muscles to mimic the action of the Achilles tendon. In some cases, a foot associated with complete rupture of the Achilles tendon dorsiflexes to a greater degree with the knee extended than the foot on the contralateral, normal side. In a prone position, the injured foot will assume a more dorsiflexed posture than on the contralateral, uninjured side.
The Thompson test most often is used to confirm the diagnosis.8 In this test, the patient lies prone on the examination table with the knee flexed and the foot hanging into dorsiflexion. The examiner squeezes the calf just distal to the widest portion of the gastrocsoleus muscle. In the presence of a normal tendon that is in continuity, the foot will plantar flex passively, bringing it upward. When the tendon is completely ruptured, the foot does not plantar flex passively; the test result is deemed positive.
O’Brien9 devised a test that he thought would have better sensitivity and specificity than the Thompson test. In this test, a small-gauge needle is injected through the skin into the tendon about 10 cm proximal to its insertion on the heel. If the tendon is intact, the needle should move when the foot is taken through a passive range of motion in dorsiflexion and plantar flexion. Lack of needle movement with ankle movement indicates that the tendon is not in continuity.
Maffulli10 prospectively investigated the relative merits of the various tests for Achilles rupture. The presence of a palpable defect was found to be the least sensitive indicator of tendon disruption. The Thompson test was found to be the most reliable way of confirming the diagnosis. In our practice, the classic history, the presence of a palpable defect, and use of the Thompson test are sufficient for making the diagnosis in the vast majority of cases.
Radiographs are not necessary to make the diagnosis in the majority of cases. The exception is in the event of a suspected distally based rupture, where a lateral radiograph may identify a bony avulsion off of the posterior calcaneus. MRI is not recommended except for circumstances in which the history and physical examination results are conflicting. Some clinicians advocate the use of ultrasonography to help guide treatment, because it may show whether the ends of the tendon can be reapproximated with plantar flexion. If ultrasonography demonstrates a residual gap between the stumps of tendon with 15° to 20° of plantar flexion, nonoperative management probably will not succeed because the tendon will be lengthened to a degree that significantly affects its function.
Treatment
Once an acute Achilles tendon rupture is identified in the emergency department/urgent care setting or primary care physician’s office, the foot should be immobilized and the patient referred promptly to an orthopedic surgeon for definitive management. Initial treatment consists of application of a well-padded short-leg plaster splint with the foot in 15° to 20° of plantar flexion to bring together the torn ends of the tendon. Theoretically, this increases the chances of successful nonoperative treatment or simplifies surgical treatment by decreasing the gap between tendon edges. If materials for a splint are not available, the foot can be immobilized in a boot with lifts placed in the heel to achieve the same effect of plantar flexion.
If surgery must be delayed beyond 1 or 2 weeks and the patient has not been immobilized correctly, a large gap may develop and is more difficult to close. This may significantly decrease the chances of success with open surgical repair and render percutaneous repair impossible.
Given the lower rate of rerupture associated with open surgical repair but a higher rate of infection and skin complications, controversy about what constitutes the optimal treatment for acute rupture of the Achilles tendon remains. The treatment choices can be categorized as 1 of 3 methods: nonoperative management, open surgical repair, or percutaneous surgical repair.
Each approach to treatment has merits and disadvantages. Nonsurgical treatment is associated with a higher rerupture rate and diminished power in the injured tendon.11,12 Open surgery of the Achilles tendon may lead to serious wound complications, including infection and skin necrosis.13 Percutaneous, or minimal, open surgical repair eliminates these risks but places the sural nerve at risk and may be associated with a higher rerupture rate than open surgery.12,14
Comparison of treatment results in studies is complicated by a lack of uniform, standardized outcome measures. A recently released clinical practice guideline from the American Academy of Orthopaedic Surgeons (adopted December 2009) states that both open and percutaneous techniques have similar outcomes and are viable surgical options.
Nonoperative treatment. The most basic form of nonoperative treatment consists of 8 weeks of cast immobilization. Initially, a splint is applied with the foot in 15° to 20° of equinus (plantar flexion) to bring the ruptured ends into apposition. When swelling goes down sufficiently, usually within 7 to 14 days, the splint is replaced with a short-leg cast in equinus or a boot with a heel lift for the remaining 6 weeks. Generally, weight bearing is allowed in the short-leg cast or boot. Crutches may be needed when weight bearing is started, but a good cast shoe or boot with a rocker bottom sole often is enough to make weight bearing less awkward. Patient adherence is mandatory.
Upon completion of the immobilization phase of treatment, the patient is instructed to begin performing gentle range of motion exercises without resistance. At the same time, the patient should wear a boot that has progressively smaller heel lifts before reverting to a regular shoe. Resistance is allowed once full range of motion is achieved. A patient should not expect to reach maximal improvement until about 12 months after the injury.
McComis and associates15 reported on 15 patients who were treated with a functional bracing regimen that allowed range of motion 2 weeks into nonoperative treatment; the study results were encouraging. Wallace and colleagues16 retrospectively evaluated the results of this protocol in 140 patients. They reported only 8 partial or complete reruptures, but only 37% of patients returned to sports activity at their preinjury level.
Some detractors of nonoperative treatment argue that the tendon’s length-tension relationship (which determines power) cannot be restored without surgically securing the tendon edges. In addition, immobilization for such a lengthy period can lead to long-term or even permanent stiffness. Indeed, most studies of nonoperative treatment show significant loss of strength and motion in the injured leg compared with the contralateral leg.12 Other authors argue that the rerupture rate, estimated at between 8% and 30%, is unacceptably high.
Open surgical repair. This comes in many forms, and there is no study that adequately shows the superiority of one method over another. In fact, most studies of operative treatment include patients who undergo a variety of surgical treatment protocols.
In operative treatment, a series of heavy, nonabsorbable sutures are passed through each stump of the tendon. The strength of the repair in tendon surgery has been shown to be directly proportional to the number of strands that cross the repair site.17 The specific configuration of these strands does not appear to affect repair strength. Some surgeons advocate the use of the plantaris tendon, gastrocnemius fascia, or allograft materials to augment the repair. However, most reserve the use of augmentation for chronic or repeated ruptures.
Surgery is best performed as soon as possible after injury, ideally within 10 days. It is considered a semi-urgent procedure, not emergent. After this period, the substance or consistency of the tendon stumps becomes weak, making the tendon less likely to hold sutures.
Bhandari and coworkers12 reported on a meta-analysis of the literature that compared operative and nonoperative management of acute rupture of the Achilles tendon. Despite a lack of homogeneity in method of surgical repair, postoperative care, and nonsurgical protocol in the 6 randomized trials that met their criteria for review, they pooled the study data. For operative and nonoperative treatment, they found average rerupture rates of 3.1% and 13% and infection rates of 4.7% and 0%, respectively. They did not identify a statistically significant difference in eventual return to function.
Percutaneous surgical repair. Ma and Griffith18 reported on 18 patients who were treated with a percutaneous method of surgical repair using 6 stab wounds to pass suture through the tendon. There was no repeated rupture, only 2 minor skin complications, and no infections. Initially, subsequent small studies of percutaneous repair seemed to show that repeated rupture is more common than with open repair.14,19
The sural nerve also is at risk for incarceration within the suture. As a result, newer methods have been developed that increase the likelihood of direct tendon end apposition and decrease the chances of sural nerve injury. To date, no study has compared percutaneous methods with open surgical methods definitively.
In our opinion, closed management is the treatment of choice for sedentary patients and those with vasculopathy or poor healing parameters. The rates of rerupture and of morbidity associated with it are unacceptably high in active persons after nonoperative treatment.
We have not had difficulty with postoperative infection. Most of our patients injure their tendon while participating in a sports or a vocational activity; they expect to return to the activity after treatment. Therefore, we offer surgical repair of the tendon to the majority of patients we see who have an acute rupture of the Achilles tendon.
Advances in limited open techniques provide improved protection for the sural nerve, prevent suture cutout, and allow for more confidence in tendon edge apposition.20 We personally have had success with a minimally invasive device in nonsedentary patients who are middle-aged or older, have mild compromise of blood flow or less than optimal skin condition, use tobacco or medications known to affect healing, or are concerned about cosmesis.
OTHER ACHILLES TENDON INJURIES
In addition to acute intrasubstance rupture, the Achilles tendon may fail at the musculotendinous junction or at its distal attachment to the calcaneus. The latter injury may be associated with avulsion of bone or may be a purely soft tissue injury.
Proximal injury
Injury to the musculotendinous junction usually occurs at the medial head of the gastrocnemius muscle. This injury often is seen in poorly conditioned men in the fourth or fifth decade of life who suddenly, forcefully, and simultaneously dorsiflex the foot and extend the knee. This maneuver occurs frequently in tennis when a player chases down a ball and quickly comes to a complete stop; therefore, it is called “tennis leg.”
On presentation, the patient reports having felt a tearing or popping sensation in the calf, followed by pain with weight bearing and weakness of dorsiflexion. In this instance, dorsiflexion via the Achilles tendon is truly preserved and the Thompson test result is not positive. Swelling, ecchymosis, and tenderness are found at the distal gastrocnemius, but there is no defect in the Achilles tendon itself. The symptoms may be confused with those of thrombophlebitis or deep venous thrombosis.
Injury to the medial head of the gastrocnemius at the musculotendinous junction may be managed symptomatically. A boot with a 2-cm heel lift that is decreased gradually over the course of 4 to 6 weeks may provide comfort during weight bearing. Surgery is not necessary. The patient should be able to return to full activities within 6 to 8 weeks.
According to many athletic trainers and physical therapists, routine calf stretching exercises may decrease the chances of gastrocnemius tear. We recommend heel cord and calf stretching exercises as part of any fitness routine to decrease the chances of injury to the musculotendinous complex and prevent strain and injury to the foot.
Distal injury
Avulsion of the tendon from its distal insertion onto the calcaneus is the least common form of acute injury to the Achilles tendon. The injury presents in 2 forms: a bony avulsion and complete soft tissue disruption. It appears to occur in an older yet active population that has had preexisting pathology at the posterior heel (eg, retrocalcaneal bursitis and exostosis, Haglund syndrome, or calcific insertional Achilles tendinitis).
The mechanism of injury that leads to tendon avulsion is similar to that of acute intratendinous rupture. However, the examiner will find ecchymosis, tenderness, and swelling on the posterior heel. The Thompson squeeze test does not elicit plantar flexion of the heel (ie, the test result usually is positive).
A bony prominence may be felt in the proximal aspect of the broad tendon insertion. If that is the case, the clinician should order a lateral radiograph of the heel. If a large avulsed bony fragment is identified, as seen in the Figure inset, the patient should be referred promptly to an orthopedic surgeon.
References:
References1. Leppilahti J, Puranen J, Orava S. Incidence of Achilles tendon rupture. Acta Orthop Scand. 1996;67:277-279.
2. Ufberg J, Harrigan RA, Cruz T, Perron AD. Orthopedic pitfalls in the ED: Achilles tendon rupture. Am J Emerg Med. 2004;22:596-600.
3. Carr AJ, Norris SH. The blood supply of the calcaneal tendon. J Bone Joint Surg. 1989;71B:100-101.
4. Aström M, Westlin N. Blood flow in the human Achilles tendon assessed by laser Doppler flowmetry. J Orthop Res. 1994;12:246-252.
5. Arner O, Lindholm A. Subcutaneous rupture of the Achilles tendon: a study of 92 cases. Acta Chir Scand Suppl. 1959;116(suppl 239):1-51.
6. Arner O, Lindholm A, Orell SR. Histologic changes in subcutaneous rupture of the Achilles tendon: a study of 74 cases. Acta Chir Scand. 1959;116:484-490.
7. Kannus P, Józsa L. Histopathological changes preceding spontaneous rupture of a tendon: a controlled study of 891 patients. J Bone Joint Surg. 1991;73A:1507-1525.
8. Thompson TC, Doherty JH. Spontaneous rupture of tendon of Achilles: a new clinical diagnostic test. J Trauma. 1962;2:126-129.
9. O’Brien T. The needle test for complete rupture of the Achilles tendon. J Bone Joint Surg. 1984;66A:1099-1101.
10. Maffulli N. The clinical diagnosis of subcutaneous tear of the Achilles tendon: a prospective study in 174 patients. Am J Sports Med. 1998;26:266-270.
11. Cetti R, Christensen SE, Ejsted R, et al. Operative versus nonoperative treatment of Achilles tendon rupture: a prospective randomized study and review of the literature. Am J Sports Med. 1993;21:791-799.
12. Bhandari M, Guyatt GH, Siddiqui F, et al. Treatment of acute Achilles tendon ruptures: a systematic overview and metaanalysis. Clin Orthop Relat Res. 2002;400:190-200.
13. Bruggeman NB, Turner NS, Dahm DL, et al. Wound complications after open Achilles tendon repair: an analysis of risk factors. Clin Orthop Relat Res. 2004;427:63-66.
14. Bradley JP, Tibone JE. Percutaneous and open surgical repairs of Achilles tendon ruptures: a comparative study. Am J Sports Med. 1990;18:188-195.
15. McComis GP, Nawoczenski DA, DeHaven KE. Functional bracing for rupture of the Achilles tendon: clinical results and analysis of ground-reaction forces and temporal data. J Bone Joint Surg. 1997;79A:1799-1808.
16. Wallace RG, Traynor IE, Kernohan WG, Eames MH. Combined conservative and orthotic management of acute ruptures of the Achilles tendon. J Bone Joint Surg. 2004;86A:1198-1202.
17. Barrie KA, Wolfe SW, Shean C, et al. A biomechanical comparison of multistrand flexor tendon repairs using an in situ testing model. J Hand Surg. 2000;25A:499-506.
18. Ma GW, Griffith TG. Percutaneous repair of acute closed ruptured achilles tendon: a new technique. Clin Orthop Relat Res. 1977;128:247-255.
19. Aracil J, Pina A, Lozano JA, et al. Percutaneous suture of Achilles tendon ruptures. Foot Ankle. 1992;13:350-351.
20. Assal M, Jung M, Stern R, et al. Limited open repair of Achilles tendon ruptures: a technique with a new instrument and findings of a prospective multicenter study. J Bone Joint Surg. 2002;84A:161-170.