Retinopathy Risk in Lupus Increases With Longer Hydroxychloroquine Use
The incidence of retinopathy among systemic lupus erythematosus (SLE) patents treated with hydroxychloroquine increased with longer treatment duration, but could be predicted by monitoring blood levels of the drug, say researchers writing in Arthritis & Rheumatology this month.
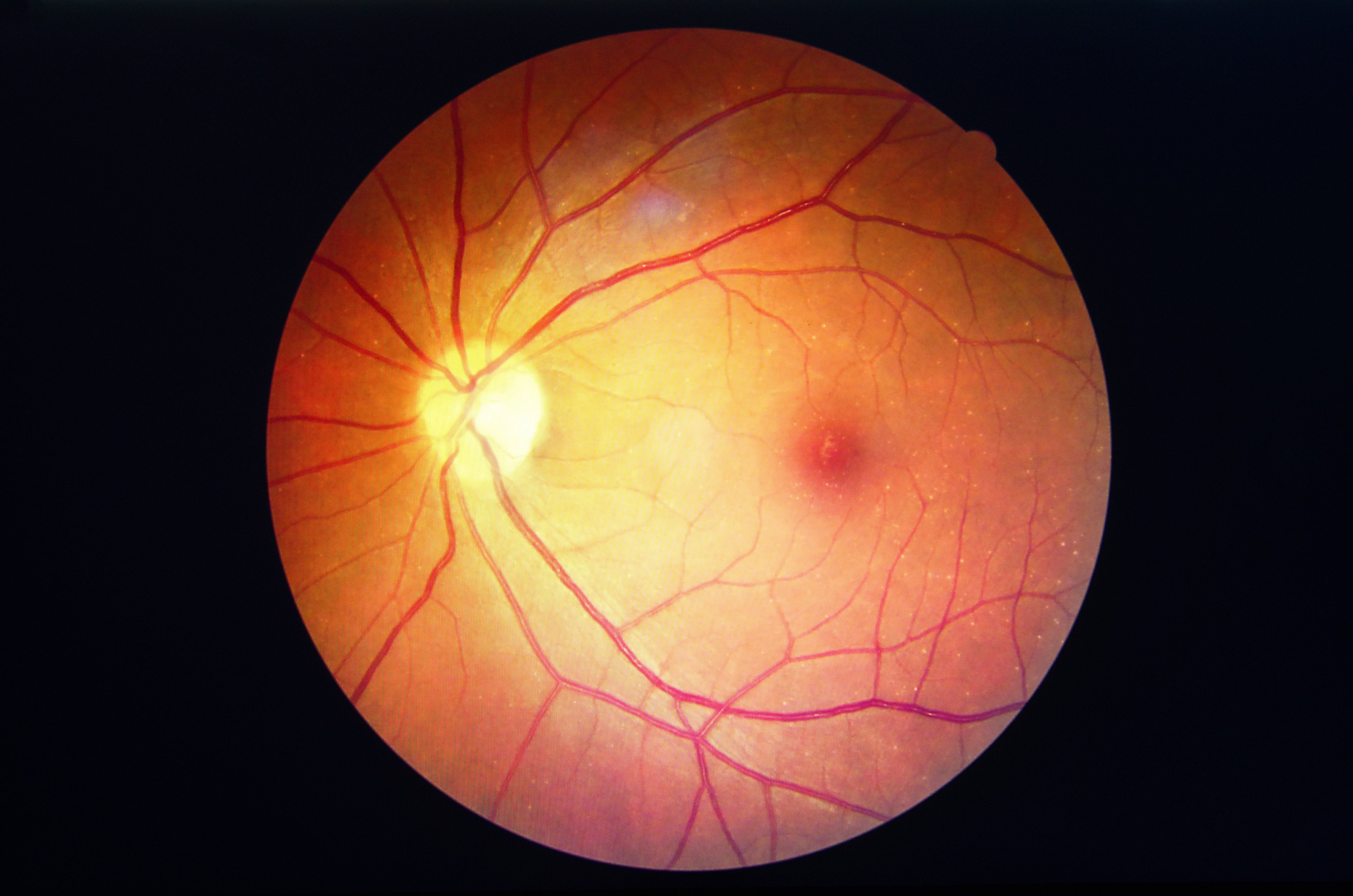
The prevalence of hydroxychloroquine retinopathy in lupus patients is much higher than previously thought, researchers report. (©AdobeStock_Koolsabuy)
The incidence of retinopathy among systemic lupus erythematosus (SLE) patents treated with hydroxychloroquine increased with longer treatment duration, but could be predicted by monitoring blood levels of the drug, say researchers writing in Arthritis & Rheumatology this month.
Hydroxychloroquine is one of only four medications approved in the U.S. for patients with systemic lupus. It is the only medication that has been shown to improve survival for patients with systemic lupus, in part, by reducing flares by as much as 50 percent. However, the treatment is associated with the development of retinopathy over time.
In this study, which was led by Michelle Petri, M.D. M.P.H., of Johns Hopkins University School of Medicine in Baltimore, researchers assessed the risk of retinopathy by examining the clinical characteristics and hydroxychloroquine levels in the blood of 537 patients with systemic lupus (494 females, 46.9 percent Caucasian and 41.5 percent African American). They found an overall frequency of retinopathy as 4.3 percent with a 1 percent risk of developing retinopathy within the first five years of treatment. The risk dropped to 1.8 percent between years six and 10; 3.3 percent between years 11 to 15 years; 11.5 percent between years 16 to 20; and, 8 percent after 21 years.
Among patients in the lowest mean tertile of hydroxychloroquine blood levels (0 to 741 ng/mL), the rate of retinopathy was 1.2 percent, 3.7 percent for those with blood levels of 741.5 to 1,176.5 ng/mL and 7.9 percent for patients with levels of 1,177 to 3,513 ng/mL (P for trend = 0.0027).
Higher toxicity rates were seen in patients with a high body mass index, patients older than 60 years (10.1 percent versus 0.5% for those more than 45 years old) and with a longer duration of treatment. African Americans had lower daily doses of hydroxychloroquine than Caucasians (4.46 versus 4.84 mg), but there were no differences in blood levels or retinopathy rates between African Americans and Caucasians. The prevalence of retinal toxicity increased in accordance with the tertile blood levels, from 1.2 percent at the lowest to 6.7 percent at the highest tertile.
"We found the results on high body mass index particularly concerning, as we capped the dose of hydroxychloroquine at 400 mg daily, no matter how high the body mass index," the authors wrote.
This is the first study that focuses on assessing retinopathy risk by tracking hydroxychloroquine blood levels.
Despite the benefits of treatment, which may also include anti-thrombotic and anti-diabetic effects and complete renal remission when given in combination with mycophenolate mofetil, the risks have been well documented. In 2016, the American Academy of Ophthalmology changed the recommended dosing of hydroxychloroquine from 6.5 mg/kg to less than 5 mg/kg, following a large retrospective study that found retinopathy rates of almost 20 percent by year 20 of use.
"Our data agree that the prevalence of hydroxychloroquine retinopathy, using newer screening technologies, is much higher than previously reported," the authors wrote, “Monitoring hydroxychloroquine blood levels is an important step to improved medication adherence in patients with SLE. We now introduce the concept of hydroxychloroquine blood level monitoring to reduce overdosage. The clinical impact of our study is that practitioners would be able to either decrease hydroxychloroquine dose or increase monitoring in patients with the highest tertile of blood levels.”
REFERENCE
Michelle Petri, Marwa Elkhalifa, Jessica Li, et al. “Hydroxychloroquine Blood Levels Predict Hydroxychloroquine Retinopathy.” Arthritis and Rheumatology. September 18, 2019. doi: 10.1002/art.41121