Current Issues in Reverse Total Shoulder Arthroplasty
A review of reverse total shoulder arthroplasty, which replaces the rotator cuff with the deltoid, often to relieve a degenerative glenohumeral. Discusses the procedure, outcomes, and complications..
ABSTRACT: Reverse total shoulder arthroplasty allows for improved function and clinical outcomes, but several issues surround its use. The chief advantage of the reverse prosthesis is its use of the deltoid as a replacement for the rotator cuff in providing elevation and abduction of the shoulder; the prosthesis is used most frequently when there is a combination of a symptomatic deficient or nonfunctional rotator cuff and a degenerative glenohumeral joint. Conditions that result in this type of pathology include rotator cuff tear arthropathy, proximal humerus fractures, and tumors. The ideal candidate for the prosthesis is a less active person older than 65 years. Clinical results may depend on the preoperative diagnosis, deltoid and teres minor function, and biomechanical design of the prosthesis. Complications include instability, neurovascular injury, and infection. (J Musculoskel Med. 2011;28:85-94)
During the past 20 years, European surgeons have used reverse total shoulder arthroplasty (RTSA) for the treatment of patients with various shoulder problems. In the United States, the FDA approved the procedure for use in patients with rotator cuff tear arthropathy in 2004.
Since its introduction, RTSA has undergone development to allow for improved function and clinical outcomes. However, several controversial issues surround the procedure. The issues involve design features of the prosthesis, the indications for surgery, the surgical approach, management of the subscapularis, and the role of a latissimus dorsi transfer to improve postoperative external rotation.
Surgeons should have a thorough understanding of shoulder anatomy and biomechanics and of the reverse prosthesis before electing to use it. Patients who may undergo the procedure should understand the inherent limitations and complications.
Both European and American surgeons have helped define the basic science and clinically relevant issues regarding the use of the RTSA prosthesis. In this article, we review the biomechanical principles, indications, clinical results, and complications of RTSA.
BIOMECHANICS
The shoulder joint consists of the humeral head and the glenoid. The design of conventional shoulder replacements depends on a functional rotator cuff to maintain the humeral component in a centered position within the glenoid. Once the humerus is allowed to migrate proximally, success after these anatomical reconstructions is diminished. For example, high rates of glenoid component loosening have been observed after conventional total shoulder replacement because the proximally migrated humeral component eccentrically loads the glenoid component.1,2
Proximal humeral head migration causes abutment against the acromial arch, renders the deltoid nonfunctional by shortening its lever arm, and contributes to persistent pain and limited forward elevation after hemiarthroplasty.3-7 Attempts at forcibly maintaining a centered humeral head with constrained and semiconstrained modifications have traded positional improvements for severely limited motion and high rates of loosening.6,8-10 As a result, a nonanatomical reverse articulation was proposed for patients with a nonfunctional rotator cuff in an effort to succeed where these anatomical designs have not.
Center of glenohumeral
rotation and deltoid function
Paul Grammont introduced the first successful reverse prosthesis in 1985. He proposed that placing the center of rotation at the prosthetic-glenoid bone interface creates less shear and lengthens the moment arm of the deltoid for improved function. The chief advantage that this device offers, like all reverse prostheses, is its use of the deltoid as a replacement for the rotator cuff in providing elevation and abduction of the shoulder.
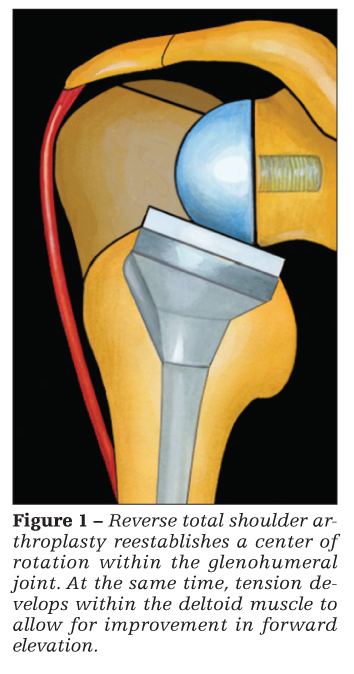
To accomplish this, the glenohumeral (GH) joint center of rotation is medialized to the level of the glenoid articular surface, which increases deltoid muscle recruitment (Figure 1).3,11,12 In addition, an increased humeral neck–shaft angle (155°) displaces the humerus inferiorly, and it improves overall deltoid function by placing the muscle on stretch and increasing its lever arm.3,11,13 Since Grammont's prosthesis was introduced, several modifications have led to the development of various types of reverse total shoulder replacement systems marketed by various companies.14
INDICATIONS
The reverse shoulder prosthesis is used most frequently in situations in which there is a combination of a symptomatic deficient or nonfunctional rotator cuff and a degenerative GH joint.15,16 Conditions that result in this type of pathology include rotator cuff tear arthropathy (GH arthritis associated with a chronic massive rotator cuff tear), proximal humerus fractures, revision total shoulder arthroplasty (TSA) with or without instability, and tumors.
In all of these cases, the rotator cuff becomes nonfunctional or irreparable. In some, the rotator cuff may be intact but severe degeneration of the posterior glenoid complicates the option of performing a traditional TSA. In these cases, an RTSA may be an option. Patients who present with a pseudoparalytic shoulder but an intact GH joint also may be candidates for RTSA to recover some function.
Chronic GH disease results in scarring, retraction, and fatty infiltration of the rotator cuff muscles.17 Proximal humerus fractures may distort the proximal humeral anatomy (tuberosities, coracohumeral arch) so severely that the rotator cuff becomes functionally useless.18
As a result, the GH joint becomes susceptible to degeneration. In these cases, where the rotator cuff does not maintain normal function, results of conventional unconstrained prostheses are suboptimal. The semiconstrained, reverse shoulder prosthesis was developed in an effort to provide a more functional alternative for patients with this type of shoulder pathology.
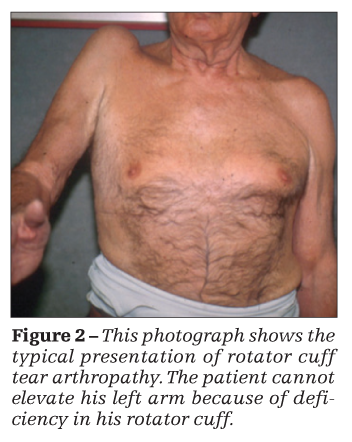
Clinically, some patients with a deficient rotator cuff and an arthritic GH joint may experience pain along with minimal or no function (Figure 2). On physical examination, they often exhibit anterosuperior escape, with forward elevation or pseudoparalysis or both. Attention must be directed toward a careful assessment of the overlying skin, deltoid muscle, and neurological function as well as bone quantity and quality of the proximal humerus and glenoid.
Ideal candidate
The ideal candidate for a reverse shoulder prosthesis is a less active person older than 65 years. Careful consideration should be given to using the prosthesis in patients who are younger than 65 years, more active (weight lifters, heavy laborers), and nonadherent to or mentally incompetent to participate in postoperative rehabilitation, because complications could result in the need for revision surgery and further compromised shoulder function.
The reverse shoulder prosthesis is used most frequently as a salvage-type solution for very disabling shoulder pathology in patients 65 years or older with low functional demand. However, in some patients (older than 65 years with greater functional demand) who have rotator cuff tear arthropathy or a proximal humerus fracture, RTSA can provide a successful clinical outcome when used as a primary procedure.
Absolute contraindications for RTSA include active infection and paralysis of the deltoid muscle or entire upper extremity. Relative contraindications include severe humeral or glenoid bone loss. Significant bone loss may occur with removal of the prosthesis in revision cases, glenoid wear, and proximal humerus fractures.
The bone available to support a reverse shoulder prosthesis needs to be evaluated in each patient. In some cases, bone grafting (autograft or allograft) techniques may be used to overcome the osseous deficit and to provide support for the prosthesis. In other cases, the bone loss may be so severe that performing an RTSA is not advised because the risk of postoperative failure is high.
CLINICAL OUTCOMES
General summary
Most of the available data on RTSA in the current literature are from retrospective (level IV) studies. The clinical results reported are mostly in patients who had a diagnosis of rotator cuff tear arthropathy. Outcomes also have been reported for patients who received the RTSA procedure for revision shoulder arthroplasty, proximal humerus fractures, osteoarthritis, and tumors.
On the basis of the results of current studies, the reverse shoulder prosthesis offers the following advantages to patients who have advanced shoulder pathology:
•Most patients show a significant improvement in pain relief.4,6,17,19-29
•Forward elevation and abduction, especially above shoulder level, improves dramatically and improves to a level that is statistically significant in most studies.3,4,6,17,18,21,23,25-28,30-32 Patients can expect postoperative forward elevation, abduction, and external rotation to be 90° to 110°, 100° to 120°, and 10° to 40°, respectively. The results in forward elevation and abduction are more consistent than in external and internal rotation. Subscapularis and teres minor integrity influence active internal and external rotation postoperatively.6,17,18,20,33-38
•Most patients define their outcome as excellent to good or at least satisfactory and improved compared with their preoperative status.28,39
Overall, clinical results may depend on the preoperative diagnosis and biomechanical design of the prosthesis. They also may depend on the integrity of the remaining rotator cuff muscles.
RTSA also has disadvantages. There is a significant complication rate, reported as high as 50% in some studies.6,34,39,40 Many complications require reoperation. The need for additional surgery in the face of advanced pathology may further compromise the patient's ability to achieve a satisfactory clinical outcome.
Although the prosthesis improves forward elevation and abduction, there may be limitations in activity. With improvement in shoulder strength and function, patients can expect to return to their daily and light recreational activities. The level of activity conducive to long-term survival of the prosthesis is unknown. In fact, there have been no definitive studies outlining the exact amount of activity, including lifting, that the reverse shoulder prosthesis can tolerate.
It seems prudent to advise patients to avoid heavy lifting and strenuous activities that place a large amount of stress across the GH joint to prevent early wear, loosening, and the need for revision surgery. However, no studies have demonstrated increased rates of complications, such as instability or loosening, in active patients. As a result, the terms “heavy” and “strenuous” provide vague descriptions for limitations in patients' activities until further research defines these issues. In looking at long-term results, Guery and associates41 reported that functional deterioration may occur as early as 6 years after surgery. More research is needed to confirm the long-term sustainability of results currently being reported for RTSA.
Outcome based on
preoperative causes
Some conclusions may be drawn from studies that compared patients who received RTSA for specific conditions. Werner and colleagues4 compared patients who received the reverse shoulder prosthesis as a primary procedure for rotator cuff tear arthropathy with ones who received it as a revision procedure after a previous shoulder surgery. Both subjective and objective values showed similar improvement in function in the groups at a minimum of 2 years after the surgery. However, there was significantly more improvement in range of motion, pain relief, and relative Constant score in patients who received the prosthesis as a primary surgery. There was no significant difference in the occurrence of complications, subjective shoulder values, or patient satisfaction between the groups. However, there was a significantly higher rate of reoperation in patients who received the reverse shoulder prosthesis as a revision procedure.4
Guery and colleagues41 reported on patients who underwent RTSA for rotator cuff tear arthropathy, rheumatoid arthritis, trauma, and revision arthroplasty. Analysis of the cumulative survival curves among these groups revealed that loosening may occur as early as 3 years postoperatively and that progressive deteriorization may occur at 6 years. At 10 years after surgery, the survival rate of the prosthesis was 91% with shoulders that had massive rotator cuff tear arthropathy, demonstrating significantly better results than those with other conditions. The survival rate of prostheses in patients with Constant scores lower than 30 was 58% at 10 years postoperatively, with no significant difference with respect to the condition.
Boileau and coworkers17 reported results from their study of a mixed population that received the reverse shoulder prosthesis. The postoperative Constant score, adjusted Constant score, and American Shoulder and Elbow Surgeons shoulder score all were significantly higher in the rotator cuff tear arthropathy group than in the revision group. The authors concluded that the results are less predictable and complication rates are higher in patients who have undergone revision surgery than in those who have rotator cuff tear arthropathy. This conclusion supports the concept that results related to RTSA may, in part, depend on the preoperative diagnosis. Overall, the results are not as good in the revision population, which has a higher rate of complications and revision surgery.
Wall and associates42 also showed that patients with posttraumatic arthritis and revision arthroplasty experience worse results than patients with rotator cuff tear arthropathy, primary osteoarthritis, and massive rotator cuff tear and those with massive rotator cuff tear alone. Although patients in the posttraumatic arthritis and revision arthroplasty groups demonstrated improvements of similar magnitude in terms of shoulder motion and function, they did not achieve the level of performance observed among patients in other groups at the most recent follow-up. Although good results are obtainable with the reverse shoulder prosthesis, the results are less predictable and the complication and revision rates are higher in revision surgery.
Survivorship
Functional results after RTSA deteriorate with time, according to a survivorship analysis performed by Favard and colleagues.32 Using cumulative survival curves, the authors determined the survival curve to complete replacement or removal, partial prosthesis replacement, reoperation with or without prosthesis replacement, and loosening; at 120 months, the global survivorship rates were 95%, 90%, 80%, and 70%, respectively. For all of these categories, there was a significantly better outcome in the patients who had a preoperative diagnosis of rotator cuff tear than in those who had revision, trauma, or arthrosis. In addition, the former group demonstrated significantly better survival at 8 years. The results were consistent with those of the previous study of Delloye and coworkers,43 which demonstrated a higher rate of mechanical complications at 6 years for revision surgery using the reverse shoulder prosthesis.
Deterioration after RTSA is multifactorial. The trend is a progressive deterioration in function after about 6 years, or earlier, depending on the diagnosis.33,38 Overall, the critical period for the prosthesis is the first 3 years after implantation. During this time, the need for most prosthetic revisions results from infection, dislocation, or rapid loosening.33,44
COMPLICATIONS
Complications frequently associated with RTSA include those of the humerus and glenoid, instability, neurovascular injury, infection, hematoma, and scapular notching.6,15,17,28,34,43,45-52 Hematoma formation is minimized by the use of drains for 24 to 72 hours postoperatively, but most hematomas resolve spontaneously.2 The risk of complications is greater when the reverse shoulder prosthesis is implanted as a revision procedure than when it is the primary operation.44 Revision surgery rates for these complications may be as high as 33%.4,17,25,31,37,52
Complications that involve the humeral side of the prosthesis include intraoperative fracture, postoperative fracture, subsidence, disassembly, radiological loosening, and the development of radiological lines.47 Those that involve the glenoid include fracture or perforation, radiolucent lines around the screws or central peg, screw fracture, disassembly of the glenosphere, and loosening.
Radiolucent lines at the base plate of the glenoid have been reported frequently, but their clinical significance has not yet been defined. Some authors have suggested that this finding may result from the offset of the glenosphere at the attachment of the base plate. Others suggest that it is caused by incomplete bone healing.17
Notching of the inferior scapula also has been reported frequently. Notching inferiorly occurs as a result of contact with the humeral component. In many cases, the notching becomes stable after 1 year, but it creates a risk of progression and potential loosening of the glenosphere.4,28
Instability, a common problem with RTSA, usually occurs within the first year after surgery. Factors related to the development of instability include the surgical approach (deltopectoral, superolateral), shortening of the subscapularis, subscapularis insufficiency, the inferior cam effect created by impingement, excessive version (anteversion or retroversion) on the humeral stem,53 and shortening of the humerus. Studies have reported instability in the range of 5% to 30%.3,4,13,25,44,53
Neurovascular complications include injury to the axillary nerve (in about 5% of cases), the suprascapular nerve, and vascular structures (in fewer than 1% of cases).54 Acromial fractures and fractures of the scapular spine are associated with osteopenia and overtensioning of the deltoid; they are rare. Walch and colleagues54 reported that infections occurred at a rate of 3% in 457 patients during the postoperative period.
CONCLUSION
Although the reverse shoulder prosthesis can provide significant improvement in clinical outcome (pain relief, forward elevation, overall satisfaction), issues remain that limit its use for anything other than a salvage-type procedure in older patients with low functional demand. Specific issues that complicate RTSA include prosthetic instability, deltoid tensioning, humeral fixation, decoaptation, scapular notching, and limitations in external and internal rotation.
The indications for RTSA are included in well-defined criteria. A shoulder surgeon with significant surgical experience is required to perform the procedure. It is clear from a review of the literature that patients who receive the reverse shoulder prosthesis as a primary procedure do better than patients who undergo the procedure as a revision surgery. However, both groups are at risk for complications.
Knowledge of the current literature helps physicians develop an understanding of the clinically relevant issues and the complications associated with the prosthesis. Future studies will allow for resolution of the controversial
issues currently surrounding RTSA and for a better understanding of how to achieve better long-term clinical outcomes.
References
1. Franklin JL, Barrett WP, Jackins SE, Matsen FA 3rd. Glenoid loosening in total shoulder arthroplasty: association with rotator cuff deficiency. J Arthroplasty. 1988;3:39-46.
2. Matsen FA 3rd, Boileau P, Walch G, et al. The reverse total shoulder arthroplasty. J Bone Joint Surg. 2007;89A:660-667.
3. Boileau P, Watkinson DJ, Hatzidakis AM, Balg F. Grammont reverse prosthesis: design, rationale, and biomechanics. J Shoulder Elbow Surg. 2005;14(1 suppl S):147S-161S.
4. Werner CM, Steinmann PA, Gilbart M, Gerber C. Treatment of painful psuedoparesis due to irreparable rotator cuff dysfunction with the Delta III reverse-ball-and-socket total shoulder prosthesis. J Bone Joint Surg. 2005;87A:1476-1486.
5. Williams GR Jr, Rockwood CA Jr. Hemiarthroplasty in rotator cuff-deficient shoulders. J Shoulder Elbow Surg. 1996;5:362-367.
6. Woodruff MJ, Cohen AP, Bradley JG. Arthroplasty of the shoulder in rheumatoid arthritis with rotator cuff dysfunction. Int Orthop. 2003;27:7-10.
7. Zuckerman JD, Scott AJ, Gallagher MA. Hemiarthroplasty for cuff tear arthropathy. J Shoulder Elbow Surg. 2000;9:169-172.
8. Lettin AW, Copeland SA, Scales JT. The Stanmore total shoulder replacement. J Bone Joint Surg. 1982;64B:47-51.
9. Post M, Haskell SS, Jablon M. Total shoulder replacement with a constrained prosthesis. J Bone Joint Surg. 1980;62A:327-335.
10. Swanson AB, de Groot Swanson G, Sattel AB, et al. Bipolar implant shoulder arthroplasty: long-term results. Clin Orthop Relat Res. 1989;249:227-247.
11. De Wilde LF, Audenaert EA, Berghs BM. Shoulder prostheses treating cuff tear arthropathy: a comparative biomechanical study. J Orthop Res. 2004;22:1222-1230.
12. Nyffeler RW, Werner CM, Gerber C. Biomechanical relevance of glenoid component positioning in the reverse Delta III total shoulder prosthesis. J Shoulder Elbow Surg. 2005;14:524-528.
13. De Wilde L, Audenaert E, Barbaix E, et al. Consequences of deltoid muscle elongation on deltoid muscle performance: a computerised study. Clin Biomech(Bristol, Avon). 2002;17:499-505.
14. Virani NA, Harman M, Li K, et al. In vitro and finite element analysis of glenoid bone/baseplate interaction in the reverse shoulder design. J Shoulder Elbow Surg. 2008;17:509-521.
15. Rockwood CA Jr. The reverse total shoulder prosthesis: the new kid on the block. J Bone Joint Surg. 2007;89A:233-235.
16. Seebauer L, Walter W, Keyl W. Reverse total shoulder arthroplasty for the treatment of defect arthropathy. Oper Orthop Traumatol. 2005;17:1-24.
17. Boileau P, Watkinson D, Hatzidakis AM, Hovorka I. Neer Award 2005: the Grammont reverse shoulder prosthesis: results in cuff tear arthritis, fracture sequelae, and revision arthroplasty. J Shoulder Elbow Surg. 2006;15:527-540.
18. Boileau P, Trojani C, Walch G, et al. Shoulder arthroplasty for treatment of sequelae of fractures of the proximal humerus. J Shoulder Elbow Surg. 2001;10:299-308.
19. Boulahia A, Edwards TB, Walch G, Baratta RV. Early results of a reverse design prosthesis in the treatment of arthritis of the shoulder in elderly patients with a large rotator cuff tear. Orthopedics. 2002;25:129-133.
20. Cuff D, Pupello D, Virani N, et al. Reverse shoulder arthroplasty for the treatment of rotator cuff deficiency. J Bone Joint Surg. 2008;90A:1244-1251.
21. De Wilde L, Mombert M, Van Petegem P, Verdonk R. Revision of shoulder replacement with a reversed shoulder prosthesis (Delta III): report of five cases. Acta Orthop Belg. 2001;67:348-353.
22. De Wilde LF, Plasschaert FS, Audenaert EA, Verdonk RC. Functional recovery after a reverse prosthesis for reconstruction of the proximal humerus in tumor surgery. Clin Orthop Relat Res. 2005;430:156-162.
23. Frankle M, Levy JC, Pupello D, et al. The reverse shoulder prosthesis for glenohumeral arthritis associated with severe rotator cuff deficiency: a minimum two-year follow-up study of sixty patients surgical technique. J Bone Joint Surg. 2006;88A(suppl 1, pt 2):178-190.
24. Frankle M, Siegal S, Pupello D, et al. The reverse shoulder prosthesis for glenohumeral arthritis associated with severe rotator cuff deficiency: a minimum two-year follow-up study of sixty patients. J Bone Joint Surg. 2005;87A:1697-1705.
25. Jacobs R, Debeer P, De Smet L. Treament of rotator cuff arthropathy with a reversed Delta shoulder prosthesis [published correction appears in Acta Orthop Belg. 2002;68:preceding 100]. Acta Orthop Belg. 2001;67:344-347.
26. Levy J, Frankle M, Mighell M, Pupello D. The use of the reverse shoulder prosthesis for the treatment of failed hemiarthroplasty for proximal humeral fracture. J Bone Joint Surg. 2007;89A:292-300.
27. McFarland EG, Sanguanjit S, Tasaki A, et al. The reverse shoulder prosthesis: a review of imaging features and complications. Skeletal Radiol. 2006;35:488-496.
28. Rittmeister M, Kerschbaumer F. Grammont reverse total shoulder arthroplasty in patients with rheumatoid arthritis and nonreconstructible rotator cuff lesions. J Shoulder Elbow Surg. 2001;10:17-22.
29. Bufquin T, Hersan A, Hubert L, Massin P. Reverse shoulder arthroplasty for the treatment of three- and four-part fractures of the proximal humerus in the elderly: a prospective review of 43 cases with a short-term follow-up. J Bone Joint Surg. 2007;89B:516-520.
30. Harman M, Frankle M, Vasey M, Banks S. Initial glenoid component fixation in “reverse” total shoulder arthroplasty: a biomechanical evaluation. J Shoulder Elbow Surg. 2005;14(1 suppl S):162S-167S.
31. Julien Y, Gondrand I, Charpenay C, et al. Shoulder reconstruction using Grammont (Delta III) total arthroplasty after resection for malignant bony tumours of proximal humerus. Eur J Orthopaed Surg Traumatol. 2003;13:77-79.
32. Favard L, Guery J, Bicknell R. Survivorship of the reverse prosthesis. In: Walch G, Boileau P, Mole D, et al, eds. Reverse Shoulder Arthroplasty. Montpellier, France: Sauramps Medical; 2006:373-379.
33. Grammont PM, Baulot E. Delta shoulder prosthesis for rotator cuff rupture. Orthopedics. 1993;16:65-68.
34. Simovitch RW, Zumstein MA, Lohri E, et al. Predictors of scapular notching in patients managed with the Delta III reverse total shoulder replacement. J Bone Joint Surg. 2007;89A:588-600.
35. Simovitch RW, Helmy N, Zumstein MA, Gerber C. Impact of fatty infiltration of the teres minor muscle on the outcome of reverse total shoulder arthroplasty. J Bone Joint Surg. 2007;89A:934-939.
36. Sirveaux F, Favard L, Oudet D, et al. Grammont inverted total shoulder arthroplasty in the treatment of glenohumeral osteoarthritis with massive rupture of the cuff: results of a multicentre study of 80 shoulders. J Bone Joint Surg. 2004;86B:388-395.
37. Sirveaux F, Favard L, Boileau P, et al. Reverse prosthesis for tumors of the proximal humerus. In: Walch G, Boileau P, Mole D, et al, eds. Reverse Shoulder Arthroplasty. Montpellier, France: Sauramps Medical; 2006:185-197.38. Renaud P, Wahab H, Bontoux L, et al. Total inverted shoulder prosthesis and rotator cuff insufficiency: evaluation and anatomical parameters predictive of good functional outcome in 21 shoulders [in French]. Ann Readapt Med Phys. 2001;44:273-280.
39. Wall B, Walch G, Jouve F. The reverse shoulder prosthesis for revision of failed total shoulder arthroplasty. In: Walch G, Boileau P, Mole D, et al, eds. Reverse Shoulder Arthroplasty. Montpellier, France: Sauramps Medical; 2006:231-242.
40. Favard L, Le Du C, Bicknell R, et al. Reverse prosthesis for cuff tear arthritis (Hamada IV and V) without previous surgery. In: Walch G, Boileau P, Mole D, et al, eds. Reverse Shoulder Arthroplasty. Montpellier, France: Sauramps Medical; 2006:113-123.
41. Guery J, Favard L, Sirveaux F, et al. Reverse shoulder arthroplasty: survivorship analysis of eighty replacements followed for five to ten years. J Bone Joint Surg. 2006;88A:1742-1747.
42. Wall B, Nov-Josserand L, O'Connor DP, et al. Reverse total shoulder arthroplasty: a review of results according to etiology. J Bone Joint Surg. 2007;89A:1476-1485.
43. Delloye C, Joris D, Colette A, et al. Mechanical complications of total shoulder inverted prosthesis [in French]. Rev Chir Orthop Reparatrice Appar Mot. 2002;88:410-414.
44. Baulot E, Chabernaud D, Grammont PM. Results of Grammont's inverted prosthesis in omarthritis associated with major cuff destruction: apropos of 16 cases [in French]. Acta Orthop Belg. 1995;61(suppl 1):112-119.
45. Bohsali KI, Wirth MA, Rockwood CA Jr. Complications of total shoulder arthroplasty. J Bone Joint Surg. 2006;88A:2279-2292.
46. Chuinard C, Trojani C, Brassart N, Boileau P. Humeral problems in reverse total shoulder arthroplasty. In: Walch G, Boileau P, Mole D, et al, eds. Reverse Shoulder Arthroplasty. Montpellier, France: Sauramps Medical; 2006:275-288.
47. Gutirrez S, Levy JC, Lee WE 3rd, et al. Center of rotation affects abduction range of motion of reverse shoulder arthroplasty. Clin Orthop Relat Res. 2007;458:78-82.
48. Gutirrez S, Greiwe RM, Frankle MA, et al. Biomechanical comparison of component position and hardware failure in the reverse shoulder prosthesis. J Shoulder Elbow Surg. 2007;16(3 suppl):S9-S12.
49. Roberts CC, Ekelund AL, Renfree KF, et al. Radiologic assessment of reverse shoulder arthroplasty. Radiographics. 2007;27:223-235.
50. Valenti PH, Bouttens D, Nerot C. Delta III reversed prosthesis for osteoarthritis with massive rotator cuff tear: long-term results (> 5 years). In: Walch G, Boileau P, Mole D, et al, eds. Shoulder Prosthesis: Two to Ten Year Follow-up. Montpellier, France: Sauramps Medical; 2001:253-259.
51. Walch G, Wall B, Mottier F. Complications and revision of the reverse prosthesis, a multicenter study of 457 cases. In: Walch G, Boileau P, Mole D, et al, eds. Reverse Shoulder Arthroplasty. Montpellier, France: Sauramps Medical; 2006:335-352.
52. Nove-Josserand L, Walch G, Wall B. Instability of the reverse prosthesis. In: Walch G, Boileau P, Mole D, et al, eds. Reverse Shoulder Arthroplasty. Montpellier, France: Sauramps Medical; 2006:247-260.
53. Gerber C. Complications and revisions of reverse total shoulder replacement. In: Walch G, Boileau P, Mole D, et al, eds. Reverse Shoulder Arthroplasty. Montpellier, France: Sauramps Medical; 2006:315-317.
54. Walch G, Wall B, Mottier F. Reversed prosthesis for primary osteoarthritis. In: Walch G, Boileau P, Mole D, et al, eds. Reverse Shoulder Arthroplasty. Montpellier, France: Sauramps Medical; 2006:149-162.