Evaluating edema of the hands
Hand edema, or “puffy hand syndrome,”is seen frequently in primary care practice, and given its broad differential diagnosis (Table), presents physicians a significant diagnostic challenge. Causes range from iatrogenic ones to end-organ disease.1
Hand edema, or “puffy hand syndrome,” is seen frequently in primary care practice, and given its broad differential diagnosis (Table A Table B), presents physicians a significant diagnostic challenge. Causes range from iatrogenic ones to end-organ disease.1 Considering the “usual suspects” of edema, such as cardiac, liver, and renal disease, is important; also considering venous thrombosis and lymphedema resulting from breast cancer treatment is a must. In addition, the history can be instrumental in ruling out the more common rheumatologic causes, such as rheumatoid arthritis (RA), gout, and pseudogout.
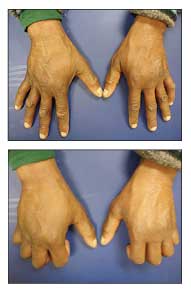
Figure 1 –A former heroin addict with hepatitis C virus infection has bilateral “puffy hands.” The hand edema begins as pitting edema, followed by thickening and fibrosis of the subcutaneous tissue.
Another common but frequently overlooked diagnosis is hepatitis C virus (HCV) infection, which often is seen in former heroin addicts whose previous habit resulted in isolated hand edema (aka “hep C hands”) (Figure 1).2 HCV infection has the highest incidence in persons born between the 1940s and the 1960s,3 and the prevalence of symptomatic HCV infection is increasing as the disease is becoming more apparent in the aging baby boomer generation.2 The “maturing process” of this population segment, dating back to the 1960s and 1970s, included widespread experimentation with drugs that ranged from LSD and other hallucinogens to hardcore intravenous (IV) heroin use. The latter resulted in repeated exposure to HCV infection (and subsequent development of chronic liver disease). Now this is an essential diagnosis to consider in such patients who have unilateral or bilateral puffy hands.
In this article, we discuss the history taking and physical examination for patients who present with puffy hands. We also describe the main clinical factors in the more common diagnoses.
HISTORY
A thorough patient history can be useful for suggesting the cause of puffy hands. Ascertaining a history of IV drug use and the location of injections is critical. Significant IV drug use resulting in chronically puffy hands strongly supports a diagnosis of HCV infection.
In addition to RA, gout, and pseudogout, the history may help rule out anasarca resulting from cardiac, renal, or liver disease. A history of infection or trauma would suggest an infectious cause or fracture; a history of autoimmune disease (eg, scleroderma or Sjgren syndrome) often is associated with hand edema.
PHYSICAL EXAMINATION
The physical examination should start with evaluation of the patient’s general appearance; the presence of scleral icterus, jaundice, and anasarca should be noted. On abdominal examination, tender hepatomegaly, splenomegaly, or lymphadenopathy may be elicited. Patients may have purpura resulting from coagulopathy or encephalopathy resulting from chronic hepatic disease. Patients also should be examined for cardiac murmurs or scars suggesting previous cardiac surgery. Renal disease may be suggested as the cause of edema by oliguria, hypertension, foamy urine, or hematuria.
Patients’ hands may suggest an association with RA with the appearance of symmetrical inflammation of the metacarpophalangeal and proximal interphalangeal joints and finger deformities, such as ulnar deviation. Erythematous, swollen, and tender joints may be related to gout or pseudogout. Evaluating whether upper extremity edema is unilateral or bilateral and whether the edema involves the whole arm or is isolated to the hand helps delineate the cause of the puffy hands.
DIFFERENTIAL DIAGNOSIS
The differential diagnosis of puffy hands is quite extensive. Many of the possible diagnoses are the ones that are most often involved (eg, congestive heart failure, nephrotic syndrome, and liver disease).
HCV infection
This is spread primarily through blood transmission, often via IV drug use (90% of cases) or blood transfusions.4 Eighty percent of HCV infection is chronic, often manifesting signs and symptoms of chronic liver disease.5 Signs include scleral icterus, jaundice, fatigue, ascites, coagulopathy, lymphadenopathy, and renal dysfunction. Portal hypertension ensues with liver failure, often resulting in encephalopathy, hepatosplenomegaly, and formation of varices because of collateral blood flow.5 Another common clinical feature of HVC infection is bilateral puffy hands.
The pathogenesis of hand edema in HVC infection has not been identified definitively. When first described by Abeles6 in 1965, this variation of hand edema was attributed to poor venous return because of thrombosis of antecubital veins after years of injected drug use. Recurrent infections at the injection site also were thought to contribute.6
Schneiderman1 suggested that the cause is lymphedema associated with the sclerosing properties of the drugs. Neviaser and colleagues7 and Foldi8 suggested that lymphatic obstruction persists after injected drug use stops, along with scarring resulting from chronic inflammation. In addition, heroin often is diluted with quinine, which is notable for destroying lymphatics.7
The hand edema begins as pitting edema, followed by thickening and fibrosis of the subcutaneous skin (see Figure 1). The lymphatics are destroyed; tissue protein increases, causing excessive connective tissue growth and resulting in fibrosis.7 Del Giudice and associates9 stated that injected drugs cause a dermal inflammatory reaction, resulting in granuloma formation similar to a foreign body–type granuloma or nonspecific granulomatous reaction.
The prevalence of HCV infection is up to 75% in injected drug users; the majority manifest chronic disease.2,4 Risk factors for puffy hands among injection drug users with HCV infection are female sex, injection into the hands, and not using a tourniquet.10 They also may include vein trauma, skin ulcers, superficial and deep venous thrombosis (DVT), and lymphatic blockage with repeated infection.9
Upper extremity thrombosis
Thrombosis of the subclavian and axillary veins results from primary or secondary causes. Primary thrombosis, known as Paget-Schroetter syndrome, occurs in healthy, young adults with strenuous exercise. The condition results from microtrauma of blood vessel intima activating the coagulation cascade; it accounts for 20% of upper extremity DVT (UEDVT).11,12
There often is an association with cervical rib abnormalities, such as thoracic outlet syndrome. This clinical syndrome causes extrinsic compression or trauma to the subclavian vein, resulting in vessel fibrosis.12 The other 80% of UEDVT cases result from secondary means, distruption of 1 or more of the components of Virchow triad: damage of blood vessel walls, hypercoagulability, and stasis.11
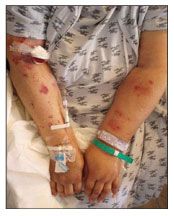
Figure 2 – Left subclavian-axillary vein thrombosis occurred in this patient after central line placement.
Thrombosis may occur in up to 72% of central venous catheter placements (Figure 2),11,13 although rates of thrombosis are rare in catheters kept in place less than 15 days.14 Hand or arm edema develops, with associated tenderness, erythema, and warmth. Pain occurs that is described as dull, constant aching; it can be disabling. Other signs include engorgement of the jugular veins and decreased shoulder joint range of motion.11,12
Lymphatic obstruction
Lymphedema results from accumulation of proteinaceous fluid in interstitial tissue; with time, there is inflammation and fibrosis of subcutaneous tissue.15,16 Slowed lymphatic transit time induces lipogenesis and fat deposition. Fibrocytes are then activated and, with time, connective tissue overgrowth occurs as fibrosis ensues (Figure 3).15
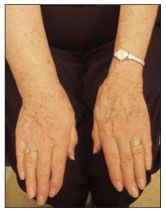
Figure 3 – Right-arm lymphedema occurred in this patient after axillary lymph node dissection.
The cause of lymphedema can be primary or secondary. Primary conditions are differentiated by age and are hereditary or spontaneous disorders of lymphatic architecture.15-18 Secondary causes are responsible for most lymphedema.
Worldwide, the most common cause of lymphedema is filariasis caused by infection with Wuchereria bancrofti, a nematode.15 In the United States, lymphedema most often is related to malignancy or treatments of malignancy.15,16 There may be upper extremity lymphatic obstruction resulting from malignancies, such as breast cancer, melanoma, and lymphoma.15 Treatment for malignancy may result in lymphedema, such as with mastectomy (24% to 49% of cases) and axillary lymph node dissection or sentinel lymph node biopsy.15,16,18
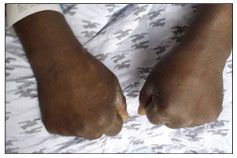
Figure 4 – Fracture of the fifth metacarpal neck resulting from trauma with a closed fist caused edema in a boxer’s hands.
Risk factors for the development of edema resulting from lymphatic disruption include trauma from previous surgeries, infection, and obesity.15,17 In addition, there is risk associated with radiation therapy causing tissue fibrosis, which constricts lymphatic channels.16 The degree of risk varies with the dose of radiation, posterior axillary boost radiation, and overlapping technique.18
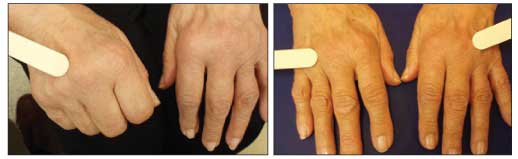
Figure 5 – Bilateral hand edema resulting from inflammation occurs in patients with rheumatoid arthritis.
Lymphedema often presents clinically as unilateral, soft, pitting edema that, over time, progresses to fibrosis. There may be pain in the affected limb with a feeling of heaviness and tightness.18 Skin manifestations include the appearance of peau d’orange (dimpling), hyperkeratosis, papillomatosis, and cellulites.15
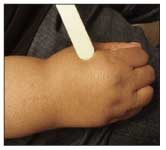
Figure 6 – In this patient, lipoma caused enlargement of the hand.
The International Society of Lymphology has described the classification of lymphedema in the following stages17:
•0: Latent or subclinical condition in which swelling is not evident despite impaired lymph transport.
•1: Mild, less than 20% increase in volume of the affected extremity; early accumulation of proteinaceous fluid subsides with limb elevation.
•2: Moderate, 20% to 40% increase in volume occurs in affected limb; limb elevation alone rarely reduces tissue swelling; pitting manifests as a result of increasing fibrosis in late stage 2.
•3: Severe, more than 40% increase in volume occurs, resulting in lymphostatic elephantiasis; there is an absence of pitting; trophic skin changes occur, including acanthosis, fat deposition, and warty overgrowth.
References:
References1. Schneiderman H. Puffy hands from drug abuse. Consultant. 2008;48:257-261.
2. Studdiford JS, Stonehouse AR, Anandanadesan R, Scott KC. Association between the “puffyhand sign” and chronic hepatitis C in a geriatric patient. J Am Geriatr Soc. 2007;55:1694-1695.
3. Armstrong GL. Injection drug users in the United States, 1979-2002: an aging population. Arch Intern Med. 2007;167:166-173.
4. Hallinan R, Byrne A, Amin J, Dore GJ. Hepatitis C virus prevalence and outcomes among injecting drug users on opioid replacement therapy. J Gastroenterol Hepatol. 2005;20:1082-1086.
5. Itskowitz MS. Hepatitis C: epidemiology, diagnosis and management. Compr Ther. 2007;33:8793.
6. Abeles H. Puffy-hand sign of drug addiction. N Engl J Med.1965;273:1167.
7. Neviaser RJ, Butterfield WC, Wieche DR. The puffy hand of drug addiction: a study of pathogenesis. J Bone Joint Surg. 1972;54A:629-633.
8. Foldi M. Diseases of Lymphatics and Lymph Circulation. Springfield, IL: Charles C. Thomas; 1969;12:30.
9. Del Giudice P, Durant J, Dellamonica P. Hand edema and acrocyanosis: “puffy hand syndrome.” Arch Dermatol. 2006;142:1084-1085.
10. Andresz V, Marcantoni N, Binder F, et al. Puffy hand syndrome due to drug addiction: a case control study of the pathogenesis. Addiction. 2006;101:1347-1351.
11. Shah MK, Burke DT, Shah SH. Upper-extremity deep vein thrombosis. South Med J. 2003;96:669-672.
12. Joffe HV, Goldhaber SZ. Upper-extremity deep vein thrombosis. Circulation. 2002;106:1874 1880.
13. Marinella MA, Kathula SK, Markert RJ. Spectrum of upper-extremity deep venous thrombosis in a community teaching hospital. Heart Lung. 2000;29:113-117.
14. Martin C, Viviand X, Saux P, Gouin F. Upper-extremity deep vein thrombosis after central vein catheterization via the axillary vein. Crit Care Med. 1999:27:2626-2629.
15. Warren AG, Brorson H, Borud LJ, Slavin SA. Lymphedema: a comprehensive review. Ann Plast Surg. 2007;59:464-472.
16. Meneses KD, McNees MP. Upper extremity lymphedema after treatment for breast cancer: a review of the literature. Ostomy Wound Manage. 2007;53:16-29.
17. International Society of Lymphology. The diagnosis and treatment of peripheral lymphedema: consensus document of the International Society of Lymphology. Lymphology. 2003;36:84-91.
18. Hinrichs CS, Watroba NL, Rezaishiraz H, et al. Lymphedema secondary to postmastectomy radiation: incidence and risk factors. Ann Surg Oncol. 2004;11:573-580.