Allopurinol Linked to Less Mortality in Patients with Gout and Cardiovascular Disease
Investigators concluded febuxostat should still be used cautiously for patients with gout and cardiovascular diseases due to inconclusive results.
In patients with gout without atherosclerotic disease, treatment with allopurinol and febuxostat showed a similar cardiovascular profile. However, treatment with allopurinol was associated with less cardiovascular mortality when compared with febuxostat in patients with a history of cardiovascular disease, according to a study published in Frontiers in Pharmacology.1
“Previous evidence showed that gout is a risk factor which can lead to cardiovascular disease,” investigators stated. “It is common that patients with gout also suffer from cardiovascular disease, and about 74% of patients have hypertension, 10% had a history of stroke, and 14% have a history of myocardial infarction… Previous meta-analysis indicated that allopurinol prevents cardiovascular disease in patients with gout; however, any potential difference in cardiovascular safety between febuxostat and allopurinol should be interpreted.”
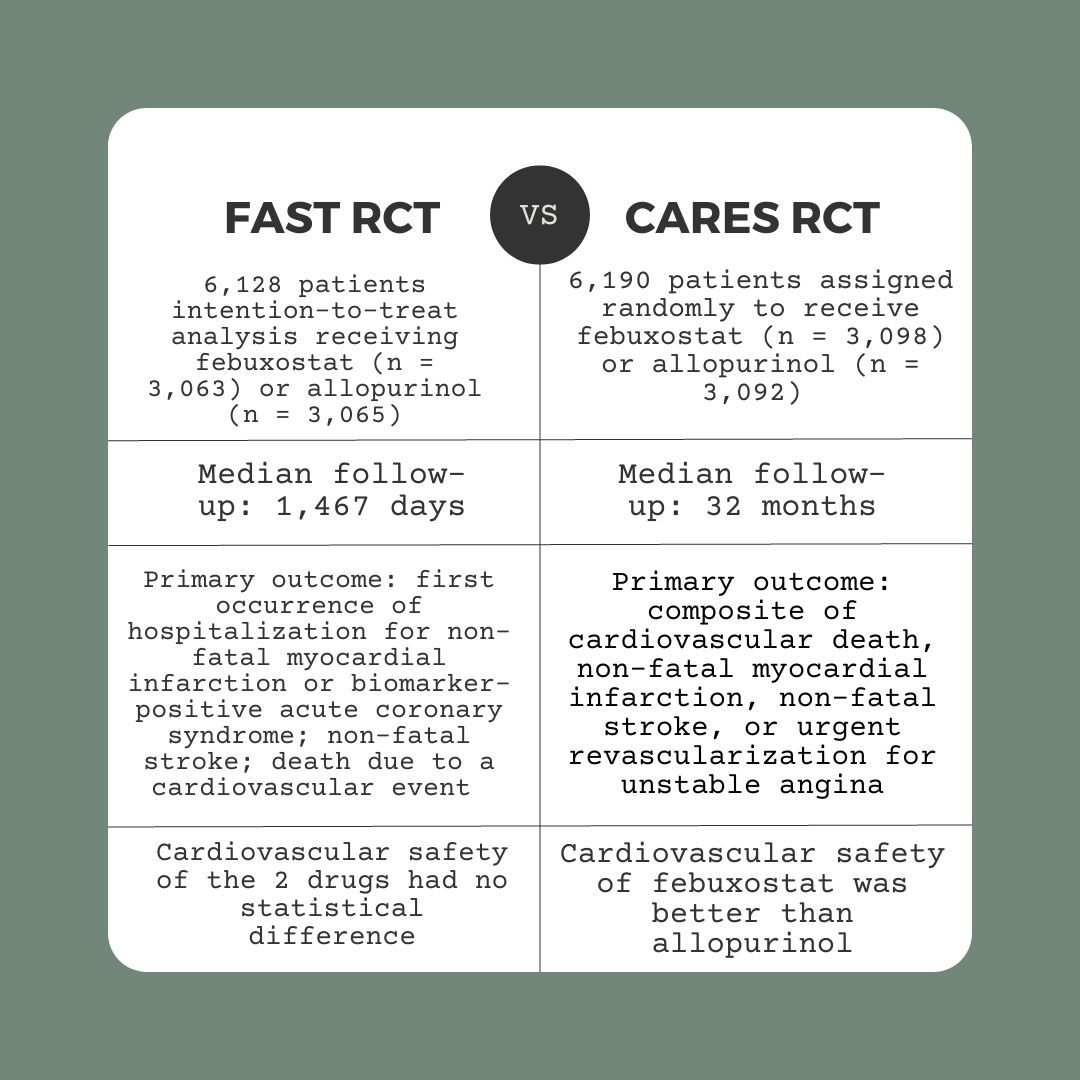
Investigators searched the databases PubMed, CBM, CNKI, Embase, WanFang, VIP, and Central from inception through January 2022 to identify randomized controlled trials (RCTs) which analyzed the cardiovascular safety of febuxostat or allopurinol treatment in adult patients with gout. Eligible studies also included major adverse cardiovascular events (MACE), cardiovascular death, all-cause death, and had a follow-up period of 52 weeks or more.
Individual patient data (IPD) were obtained and reconstructed based on the Kaplan-Meier curves in 2 studies (the febuxostat versus allopurinol streamlined trial [FAST] and the cardiovascular safety of febuxostat and allopurinol in patients with gout and cardiovascular morbidities [CARES] trial). Time-varying risk ratios (RRs) summarized time-to-event outcomes. A multi-level flexible hazard regression model calculated RRs of MACE incidence, cardiovascular mortality, and all-cause mortality. Two-stage meta-analyses analyzed the quantitative estimates of time-specific relative risks at 6 time points (1 , 2, 3, 4, 5, and 6 years) using reconstructed IPD.
The 2 RCTs selected evaluated 12,318 participants. There was no significant difference in the incidence of MACE between both groups (RR = 0.99 [95% CI, 0.89–1.11], p = 0.87). Further, there was no significant difference in both cardiovascular mortality (RR = 1.17 [95% CI, 0.98–1.40], p = 0.08) or all-cause mortality (RR = 1.03 [95% CI, 0.91–1.17], p = 0.62). No significant difference was reported in any outcomes at any of the time points in the 2-stage meta-analyses.
The study was limited by the strict inclusion criteria, such as only using studies that were published in either Chinese or English. However, investigators believe the results can still provide clinical references for treatment of gout as these patients often have comorbid diseases. Another limitation was that only 2 studies were included; therefore, future relevant RCTs or real-world studies are needed.
“Febuxostat likely has a similar cardiovascular profile to allopurinol in patients without atherosclerotic disease based on the reconstructed IPD,” investigators concluded. “However, in patients with a history of cardiovascular disease, allopurinol treatment is associated with less cardiovascular mortality as compared with febuxostat. Because their results are inconclusive, febuxostat still needs to be used cautiously for patients with gout and cardiovascular diseases.”
Reference:
Guan X, Zhang S, Liu J, et al. Cardiovascular safety of febuxostat and allopurinol in patients with gout: A meta-analysis. Front Pharmacol. 2022;13:998441. Published 2022 Sep 30. doi:10.3389/fphar.2022.998441