Phase 2B Trial of Tildrakizumab Shows Promise in Psoriatic Arthritis
A 24-week trial of tildrakizumab to treat the skin and joint conditions associated with psoriatic arthritis shows promise as a possible new treatment, shows a study presented at EULAR.
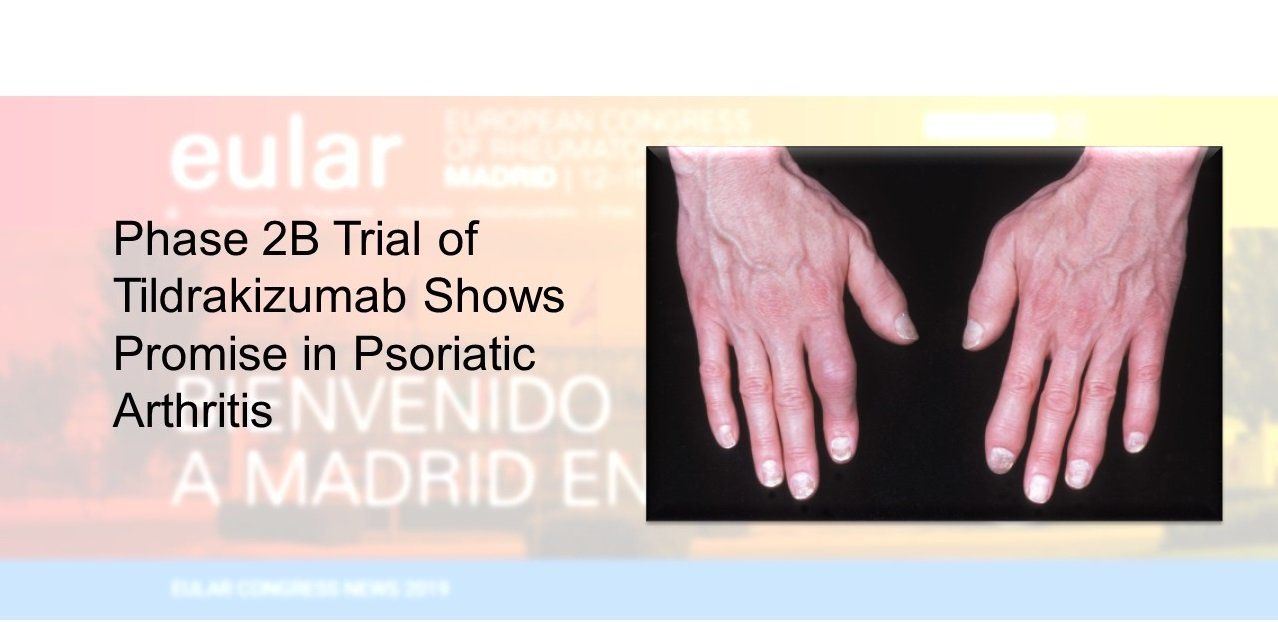
A 24-week trial of tildrakizumab to treat the skin and joint conditions associated with psoriatic arthritis shows promise as a possible new treatment. (MedicalIllustrations.com, 01B0W1EW)
A 24-week trial of tildrakizumab to treat the skin and joint conditions associated with psoriatic arthritis shows promise as a possible new treatment, shows a study presented at the European Congress of Rheumatology (EULAR) annual meeting this weekend in Madrid.
The phase two B trial, which was led by Philip J. Mease, M.D., Swedish Medical Center/Providence St. Joseph Health and the University of Washington, showed a clear separation between the treatment and placebo groups by eight weeks in order to achieve ACR20. ACR50 was achieved by 12 weeks.
“Our results demonstrate a clear separation between tildrakizumab and placebo as early as eight weeks,” Dr. Mease said. “A promising role is suggested for tildrakizumab in the treatment of patients suffering with psoriatic arthritis.”
Tildrakizumab is an IL-23p19 monoclonal antibody currently approved to treat moderate-to-severe plaque psoriasis. Currently, patients with peripheral arthritis from psoriatic arthritis are treated first with synthetic DMARDs. Patients with refractory disease are then treated with biologic DMARDs targeting TNF, IL-17 or IL12/23.
This was a four-week randomized, double-blind, placebo-controlled, multiple-dose, phase two B trial of 391 patients with psoriatic arthritis of at least six months and who had placebo-controlled, multiple-dose, phase 2B study. It included 391 adults with psoriatic arthritis who had three or more tender or swollen joints. They were randomized to receive tildrakizumab 200 mg once every four weeks, 200 mg, 100 mg, 20 mg every 12 weeks, or placebo every four weeks.
By week 24, a significantly higher proportion of patients receiving tildrakizumab at any dose achieved a 90 percent reduction in Psoriasis Area and Severity Index (PASI 90), and a 50 percent reduction in American College of Rheumatology response criteria (ACR 50) versus placebo.
There were four active treatment groups in which patients received 20 mg, 100 mg or 200 mg tildrakizumab every 12 weeks, or 200 mg every four weeks. There was a dose response with improvement with increased dosage. However, the shortening of dosing interval of 200 mg from 12 to four weeks did not result in a measurable increase in skin or joint response scores.
In patients receiving 200 mg tildrakizumab every 12 weeks, 79.6 percent and 50 percent achieved PASI 75 and PASI 90 respectively versus 16.7% and 7.1% in the placebo group (p<0.0001).
“We welcome these promising results for tildrakizumab in patients with psoriatic arthritis,” said Hans Bijlsma, M.D., Ph.D., EULAR president. “Extending research into different patient groups may bring benefits that address current unmet needs.”
DISCLOSURES:
The authors reported a number of disclosures with pharma.
REFERENCE:
Mease PJ, Chohan S, Garcia Fructuoso FJ, et al. “Randomized, double-blind, placebo-controlled, multiple-dose, phase 2b study to demonstrate the safety and efficacy of tildrakizumab, a high-affinity anti–interleukin-23p19 monoclonal antibody, in patients with active psoriatic arthritis.” EULAR 2019; Madrid: Abstract LB0002.