RediTrex: Innovative Subcutaneous Injection Administration of Methotrexate
Theresa Human, PharmD, discusses RediTrex, a subcutaneous injection method of administering methotrexate.
Rheumatology Network interviewed Theresa Human, PharmD, to discuss RediTrex, a subcutaneous injection method of administering methotrexate. Human is the Medical Science Liaison for Cumberland Pharmeceuticals. She explains what sets RediTrex apart from other treatment methods, the clinical significance, and the many benefits of receiving RediTrex as opposed to oral methotrexate for certain patient populations.
Rheumatology Network: What initially inspired the team to begin creating a subcutaneous injection method of administering methotrexate rather than the traditional oral version?
Theresa Human, PharmD: We know from the literature that not everyone is able to tolerate the doses of oral methotrexate. And giving a subcutaneous is really an alternative that's been available for quite some time in patients who need doses greater than 15 to 20 milligrams and those who are unable to tolerate the oral versions. The bioavailability isn't always perfect in these patients. And so sometimes the subcutaneous is just more effective. We know methotrexate is definitely the drug of choice for many of the disease states that we see methotrexate used in the outpatient setting and subcutaneous just seems to be more effective, especially at those higher doses. So, this new delivery device was created by a multinational team over in Europe. And really with the patient, the end user, in mind, dexterity can become an issue in some of these patients where they're unable to use the vial and put the needle into the vial, withdrawal the correct contents, etc. It has a larger flange so that those with dexterity issues may be able to hold the syringe a little bit better. It has a 5 bevel needles so that it's less painless whenever you're administering the agent and a smaller needle as well. So, I think a lot of all these things culminate into just a nice delivery device for these patients.
RediTrex device
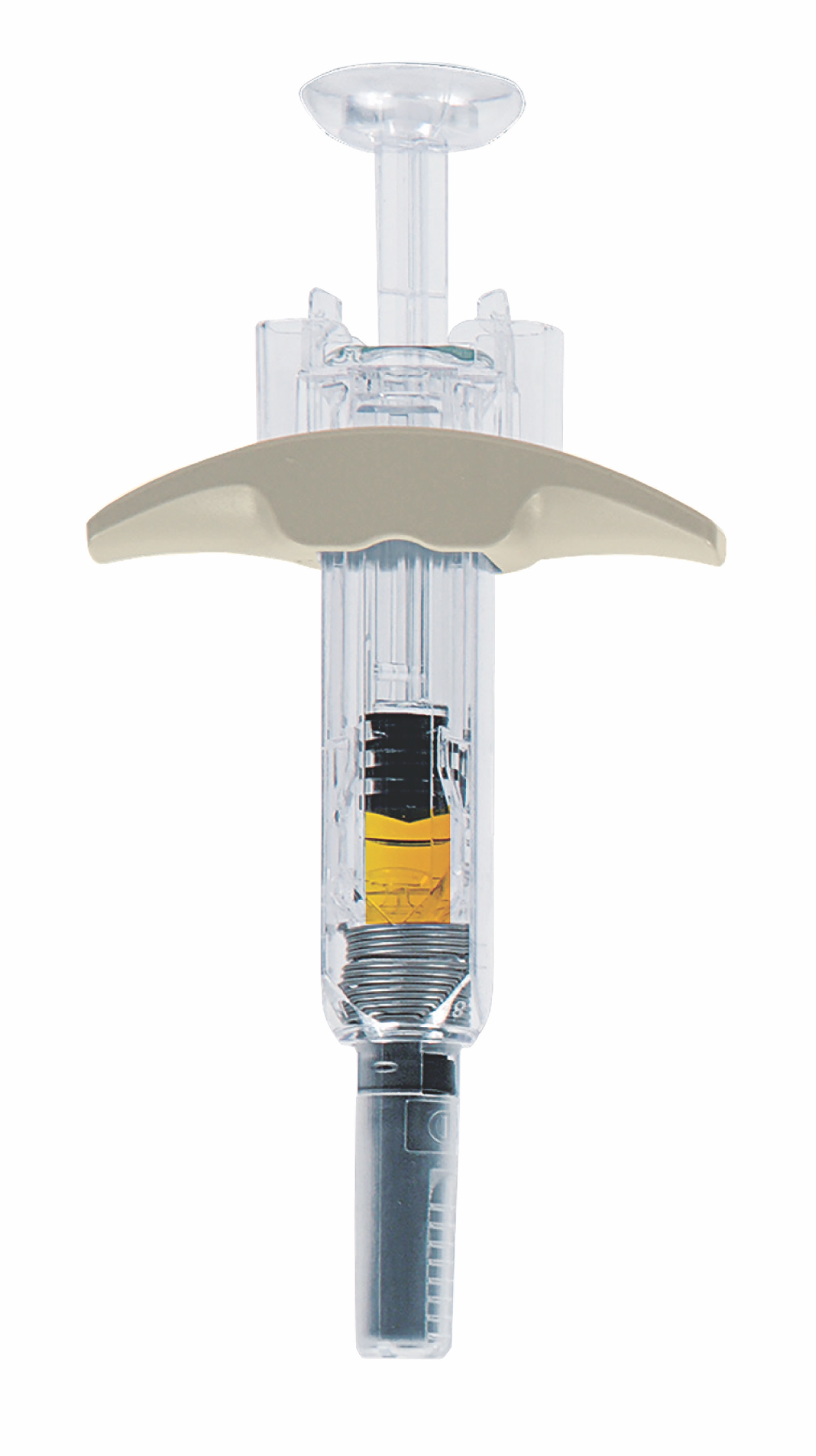
RN: What sets RediTrex apart from other treatment methods on the market?
TH: That's a great question as well. The forms that are out there currently are the oral tablets. There's also a liquid methotrexate for the pediatric indications and those patients that are able to tolerate first line with oral therapy. There are then several different subcutaneous ones available. So, we have our prefilled syring and there are a couple of auto injectors as well. Auto injectors are great, but they're a little bit more expensive. Some individuals are unable to tolerate the high rate of injection that occurs and it can become painful. So, in that case, these prefilled syringes come in the dose that you need, you're able to inject it as slowly or as quickly as you want, and therefore hopefully reduce the pain that can sometimes be felt.
RN: What conditions can RediTrex treat?
TH: We are indicated currently for idiopathic polyarticular juvenile idiopathic arthritis, psoriasis, and rheumatoid arthritis.
RN: What's the clinical significance of the FDA approval?
TH: The well so methotrexate has been around forever. So, we don't really need the new drug indication. Of course, I think it's really the delivery system that RediTrex gives the individuals, the end users, and the patients. Just as I mentioned before, it's very easy to use. It has the end where you have the large flange and the concave no-slip administration device. And again, a lot of these patients end up having dexterity issues, coordination issues, and they're unable to use some of their fingers like they normally would be able to earlier, so I really think it all has to do with the administration availability and that the syringe was designed specifically for this patient population. So, it really isn't so much the methotrexate because everyone knows methotrexate and where it's supposed to go in these specific devices or in these specific indications. But really, it's all in the delivery device.
RN: Does the team plan on doing any further research on this topic?
TH: Oh, I'm sure we would certainly love to do lots of research on the topic, as we’ve only had the product now for a little while, I think we're still trying to figure out how to get it to the right customer and really kind of look at those delivery devices and understand better what population it might go best with.
RN: Is there anything else that you would like our audience to know before we wrap up?
TH: Being a clinical sciences scientist, cost isn't always a big thing that I want to speak to. But I do think for the end users, especially in this day and age where everything's very expensive and not everything is covered, I do think cost is also a thing that we should probably bring out there. And we do know that it's half the cost of the auto injectors at the wholesale acquisition price, which for some people could mean that it's a better option for them. I do need to mention that I'm not saying that auto injectors are bad at all, because I think they're fantastic. They're another tool in our toolbox for treating these patients with arthritis, and polyarticular arthritis, and psoriasis, but [RediTrex] just gives us another tool to use.