Woman With Sarcoidosis, Lymphoma, and Shoulder Pain
A 56-year-old woman presents with a history of intermittent shoulder pain for the past six months. Early signs point to bursitis, but the plot thickens: She also has sarcoidosis and lymphoma. How would you proceed?
A 56-year-old African American female presented to the outpatient clinic with a six-month history of left shoulder pain. In 2005, she had been diagnosed with sarcoidosis of the lungs and liver, based on on transbronchial and liver biopsies showing epithelioid non-necrotizing granulomas, as well as with cutaneous T-cell lymphoma diagnosed by skin biopsy from the scalp.
She described her shoulder pain as 5/10 in intensity, aching to sharp, mainly on the anterior and lateral aspect of the shoulder but radiating to the arm. The pain was intermittent, relieved by rest and aggravated by movement. Range of motion was decreased compared to the contralateral side, and she was unable to actively abduct her left arm above 90 degrees secondary to pain, which also reduced the extremes of motion by about 20 degrees on all planes.
She denied any history of trauma, neck or back pain, numbness, tingling, or paresthesias in the distal extremity. For the past 2 years, she had been taking prednisone 5 mg daily along with pain medication.
On physical examination, she appeared in mild distress due to joint pain. Her vital signs were unremarkable except for mild hypertension (149/88) and BMI of 34. The erythrocyte sedimentation rate was 50 mm/hr. Other laboratory findings (angiotensin-converting enzyme, antinuclear antibody, and hepatitis A, B, and C antibodies) were all negative.
Laboratory findings showed an erythrocyte sedimentation rate (ESR) of 50 mm/hr. Levels of angiotensin-converting enzyme, antinuclear antibody and Hepatitis A, B and C antibodies were all within the normal range.
Which imaging tests would you order to diagnose this shoulder pain?
For the answer, go to the next page.
Imaging studies including plain X-ray and a computed tomography (CT) scan of the shoulder were negative, and a nuclear scan was negative for any uptake.
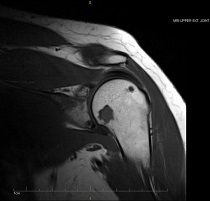
MRI of the left shoulder joint with and without contrast (left) was performed, revealing multiple enhancing lesions in the glenoid and the medial aspect of the metaphysis of the left humerus. Bone scans revealed symmetric increased radiotracer uptake in both shoulders, hips and knees.
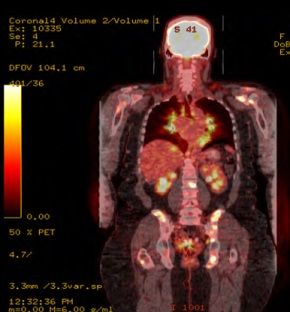
FDG-Positron emission tomography (PET) scans showed uptake in the lymph nodes involving the hila, mediastinum and retroperitoneum, along with diffusely increased uptake of the left hepatic lobe. Multiple foci of increased FDG uptake involving both axial and proximal appendicular skeleton were seen as well, consistent with either sarcoidosis or lymphoma.
What diagnosis do you suspect for this finding on FDG-PET? Which further tests would you order? See the diagnosis, images, and discussion.
Diagnosis: Sarcoidosis of the humerus
CASE DESCRIPTION:
Clinically, the patient showed the following signs:
• Diffusely tender to palpation across the anterior aspect of the left shoulder and at the deltoid muscle.
• Pinpoint tenderness over the greater tubercle, over the anterior aspect of her shoulder as well as over the pectoralis muscle. • Tenderness to palpation over the AC joint.
• Unable to actively abduct her left arm above 90 degrees, secondary to pain, with decreased. range of motion when compared to contralateral side. Good range of forward flexion, adduction, and internal and external rotation, except at the extremes of motion she was lacking about 20 degrees in all planes, limited by pain.

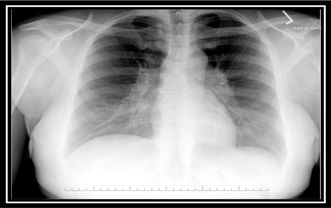
During the initial evaluation, a chest x-ray (left) revealed reticulonodular interstitial disease in the lower lobes, widening of the mediastinum and mediastinal lymphadenopathy.
An X-ray of the shoulder (below, right) revealed only mild
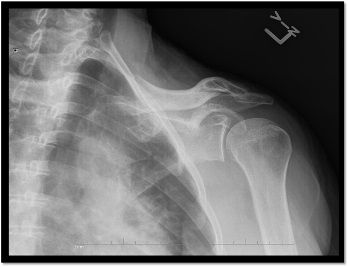
acromioclavicular and glenohumeral joint osteoarthritis. The working diagnosis was rotator cuff tendinitis versus subacromial bursitis.
A lidocaine injection, to determine whether the pain was originating from soft tissue or from the glenohumeral joint, did not improve her symptoms. This triggered further investigations.
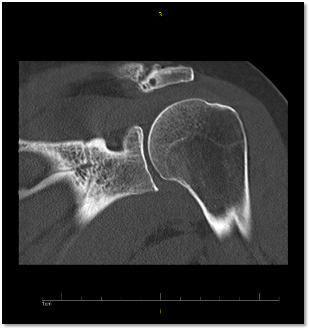
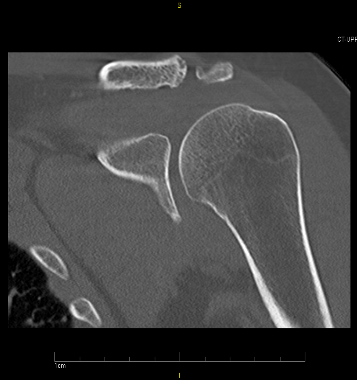
CT without contrast revealed no focal lytic or blastic lesions, showing only mildacromioclavicular joint osteoarthritis with osteophyte formation, subchondral cystic changes and sclerosis (above).
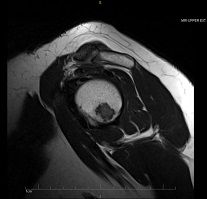
Subsequent MRI (below) showed multiple enhancing lesions in the glenoid and the medial aspect of the metaphysis of the left humerus, as well as tenosynovitis of the long head of the biceps tendon and, again, osteoarthritis of the acromioclavicular joint with effacement suggestive of subdeltoid bursitis, as well as mild rotator cuff tendinopathy.

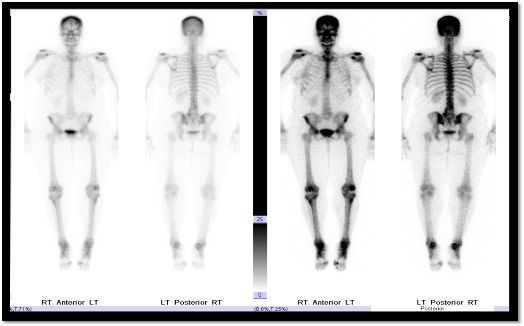
A bone scan revealed no abnormal accumulation of radiotracer, and thus was not helpful in ruling out metastatic bone lesions. The symmetrically increased radiotracer uptake of both shoulders, hips, and knees were likely degenerative.
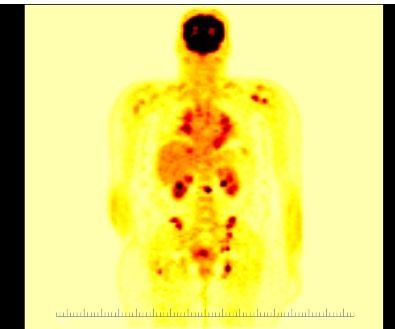
FDG PET revealed increased FDG uptake involving both axial and proximal appendicular skeleton (below), possibly related to either sarcoidosis or lymphoma. Pathological testing was essential to making the correct diagnosis.
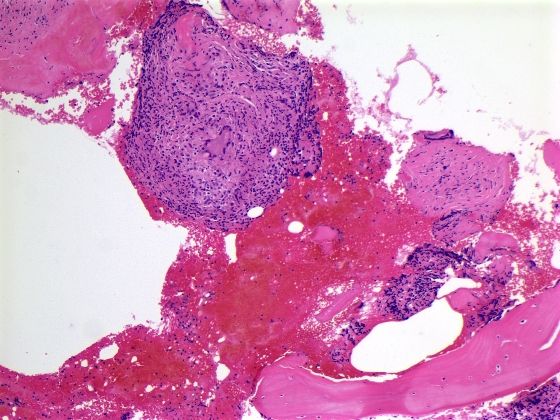
Six biopsies of humeral head and left glenoid stained with hematoxylin (blue) and eosin (pink) showed multiple noncaseating granulomas.
This pathological picture can be seen in tuberculosis and fungal infections such as histoplasmosis, which were ruled out by doing special stains for acid fast and fungal stains. These were negative.
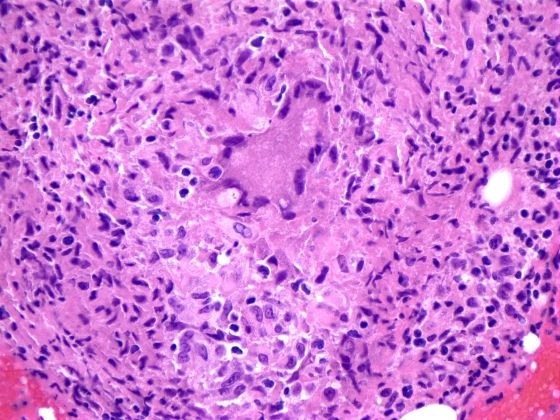
As noncaseating granulomas can also be seen in lymphoma, it remained important to rule out metastasis. Immunohistochemistry was negative for CD20, CD3, CD79a, CD15, CD30, EBV-LMP, CD45RO, EMA, and BCL-2. Accordingly, the diagnosis of sarcoidosis was made.
DISCUSSION
While osseous sarcoid has been reported in a variety of other bones, there are few reported cases involving the humerus. There are no case reports describing humeral involvement in a patient with sarcodiosis and concomitant cutaneous T-cell lymphoma.
Sarcoidosis is a systemic inflammatory disorder of unknown etiology. It is characterized pathologically by noncaseating epithelioid cell granulomas, primarily affecting the lungs and the lymphatics, with variable manifestations and course.1 Other organ involvement can include the skin, kidneys, liver, salivary glands, and bone marrow.
Musculoskeletal involvement is infrequent. While an acute, self-limited polyarthritis may be observed in up to 40% of sarcoid patients, other manifestations such as chronic arthritis (1-4%) or symptomatic muscle involvements (< 0.5%) are rare.2
The diagnosis of sarcoidosis is made by a combination of clinical, radiological, and histological findings. However, histological proof with noncaseating granulomata remains the hallmark of diagnosing this disease. Sarcoidosis may simulate many diseases, including metastatic malignancies, making differential diagnosis a challenge.
The frequency of bone involvement in sarcoid varies from 1% to 13%, with an average of 5%.3-4 The overall prevalence of clinically significant bone involvement in sarcoidosis is low (2 – 5%).2 Granulomatous bone involvement has an overall incidence of 1–13%, which is probably underestimated in some patient series because bone involvement is often asymptomatic.5 Shor et.al reported the incidence of skeletal involvement with sarcoidosis to range from 3% to as much as 36% in patients with musculoskeletal complaints.6 Bone lesions occur most frequently in the phalanges of the fingers and toes, and less often in the skull, vertebrae, and pelvis.7
The relatively rare presentation of osseous sarcoidosis can make radiographic diagnosis difficult, and probably results in a substantial underdiagnosis of sarcoid bone disease when it is mistaken for tendinitis or bursitis. X-ray evaluation of the skeleton often shows normal findings but can reveal focal osteosclerosis and osteolysis. CT of the skeleton may show focal sclerotic lesions. MRI is sensitive (45%) for detection of osseous sarcoidosis, but is nonspecific.8
In our case, initially the patient was thought to have disseminated metastatic malignancy, given the history of cutaneous T-cell lymphoma. Extracutaneous manifestations of T-cell lymphoma include involvement of regional lymph nodes (approximately 30% in mycosis fungoides),9 lungs,10 spleen, liver, and gastrointestinal tract. Bone marrow involvement is rare.11
(Scroll beyond the Comment box below to see References.)
References:
REFERENCES
1. Spagnolo P, Luppi F, Roversi P et al. Sarcoidosis: Challenging Diagnostic Aspects of an Old Disease. Am J Med. (2012) 125:118-125.
2. Zisman DA, Shorr AF, Lynch JP, 3rd. Sarcoidosis involving the musculoskeletal system. Semin Respir Crit Care Med (2002) 23:555-570.
3. Wilcox A, Bharadwaj P, Sharma OP. Bone sarcoidosis. Curr Opin Rheumatol (2000) 12:321-330.
4. Beasley EW, 3rd, Peterman SB, Hertzler GL. An unusual form of tibial sarcoidosis. AJR Am J Roentgenol (1987) 149:754-756.
5. Bargagli E, Olivieri C, Penza F et al Rare localizations of bone sarcoidosis: two case reports and review of the literature. Rheumatol Int. (2011) 31:1503-1506.
6. Shorr AF, Murphy FT, Gilliland WR et al. Osseous disease in patients with pulmonary sarcoidosis and musculoskeletal symptoms. Respir Med (2000)94:228–232.
7. Uehlinger E and Wurm K. Sarcoidosis of the skeleton: review of the literature and case report. ROFO Fortschr Geb Rontgenstr Nuklearmed (1976)125:111–122
8. Moore SL, Teirstein A, and Golimbu C. MRI of sarcoidosis patients with musculoskeletal symptoms. AJR Am J Roentgenol (2005) 185:154-159.
9. Vonderheid EC, Diamond LW, van Vloten WA et al. Lymph node classification systems in cutaneous T-cell lymphoma. Evidence for the utility of the Working Formulation of Non-Hodgkin's Lymphomas for Clinical Usage. Cancer (1994) 73:207.
10. Baser S, Onn A, Lin E et al. Pulmonary manifestations in patients with cutaneous T-cell lymphomas. Cancer (2007) 109:1550
11. World Health Organization Classification of Tumours, Pathology and Genetics of Tumours of Haematopoietic and Lymphoid Tissues, Jaffe ES, Harris NL, Stein H, Vardiman JW (Eds), IARC Press, Lyon 2001.